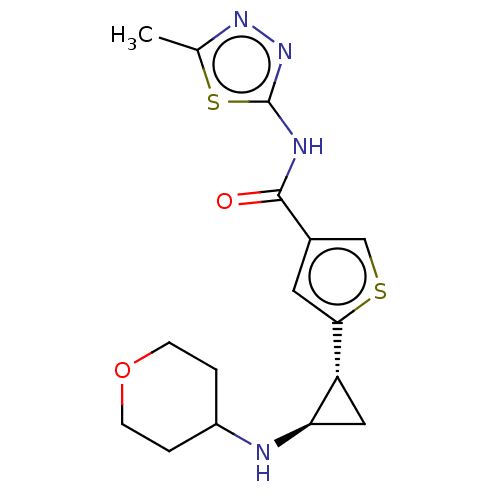
BDBM243415 N-(5-methyl-1,3,4- thiadiazol-2-yl)-5- ((1R,2R)-2- (tetrahydro-2H- pyran-4-ylamino)- cyclopropyl)thio- phene-3-carboxamide::US10053456, 49::US10414761, Example 49::US10968213, Example 50::US9718814, 49::US9920047, 49
SMILES Cc1nnc(NC(=O)c2csc(c2)[C@@H]2C[C@H]2NC2CCOCC2)s1
InChI Key InChIKey=NELDJPIPYKJYET-CHWSQXEVSA-N
Data 21 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 21 hits for monomerid = 243415
Found 21 hits for monomerid = 243415
Affinity DataIC50: <100nMpH: 8.0 T: 2°CAssay Description:A test compound dissolved in DMSO was added by to a reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing LSD1 enzyme, and the m...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:The MAO-B inhibitory activity evaluation described below followed the protocol of MAO-Glo (registered trademark) Assay of Promega KK.A test compound ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMpH: 7.5 T: 2°CAssay Description:A test compound dissolved in DMSO was added to a reaction solution (100 mM HEPES (pH 7.5), 5% glycerol, 10% DMSO) containing MAO-B enzyme (Sigma-Aldr...More data for this Ligand-Target Pair
Affinity DataIC50: <100nMpH: 8.0 T: 2°CAssay Description:A test compound dissolved in DMSO was added by to a reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing LSD1 enzyme, and the m...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMpH: 7.5 T: 2°CAssay Description:The MAO-A inhibitory activity evaluation described below followed the protocol of MAO-Glo (registered trademark) Assay of Promega KK.A test compound ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMpH: 7.5 T: 2°CAssay Description:The MAO-B inhibitory activity evaluation described below followed the protocol of MAO-Glo (registered trademark) Assay of Promega KK.A test compound ...More data for this Ligand-Target Pair
Affinity DataIC50: <100nMpH: 8.0 T: 2°CAssay Description:A test compound dissolved in DMSO was added by to a reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing LSD1 enzyme, and the m...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMpH: 7.5 T: 2°CAssay Description:The MAO-A inhibitory activity evaluation described below followed the protocol of MAO-Glo (registered trademark) Assay of Promega KK.A test compound ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMpH: 7.5 T: 2°CAssay Description:The MAO-B inhibitory activity evaluation described below followed the protocol of MAO-Glo (registered trademark) Assay of Promega KK.A test compound ...More data for this Ligand-Target Pair
Affinity DataIC50: <100nMAssay Description:A test compound dissolved in DMSO was added by to a reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing LSD1 enzyme, and the m...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:A test compound dissolved in DMSO was added to a reaction solution (100 mM HEPES (pH 7.5), 5% glycerol) containing MAO-A enzyme (Sigma-Aldrich Co. LL...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:A test compound dissolved in DMSO was added to a reaction solution (100 mM HEPES (pH 7.5), 5% glycerol, 10% DMSO) containing MAO-B enzyme (Sigma-Aldr...More data for this Ligand-Target Pair
Affinity DataIC50: <100nMAssay Description:LSD1: A test compound dissolved in DMSO was added by to a reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing LSD1 enzyme, and...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:MAO-A: The MAO-A inhibitory activity evaluation described below followed the protocol of MAO-Glo (registered trademark) Assay of Promega KK.A test co...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:MAO-B: The MAO-B inhibitory activity evaluation described below followed the protocol of MAO-Glo (registered trademark) Assay of Promega KK.A test co...More data for this Ligand-Target Pair
Affinity DataIC50: <100nMAssay Description:LSD1: A test compound dissolved in DMSO was added by to a reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing LSD1 enzyme, and...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:The MAO-A inhibitory activity evaluation described below followed the protocol of MAO-Glo (registered trademark) Assay of Promega KK.A test compound ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:The MAO-B inhibitory activity evaluation described below followed the protocol of MAO-Glo (registered trademark) Assay of Promega KK.A test compound ...More data for this Ligand-Target Pair
Affinity DataIC50: <100nMAssay Description:LSD1: A test compound dissolved in DMSO was added by to a reaction solution (50 mM Tris-HCl (pH 8.0), 0.1% BSA, 1 mM DTT) containing LSD1 enzyme, and...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:The MAO-A inhibitory activity evaluation described below followed the protocol of MAO-Glo (registered trademark) Assay of Promega KK.A test compound ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMpH: 7.5 T: 2°CAssay Description:A test compound dissolved in DMSO was added to a reaction solution (100 mM HEPES (pH 7.5), 5% glycerol) containing MAO-A enzyme (Sigma-Aldrich Co. LL...More data for this Ligand-Target Pair