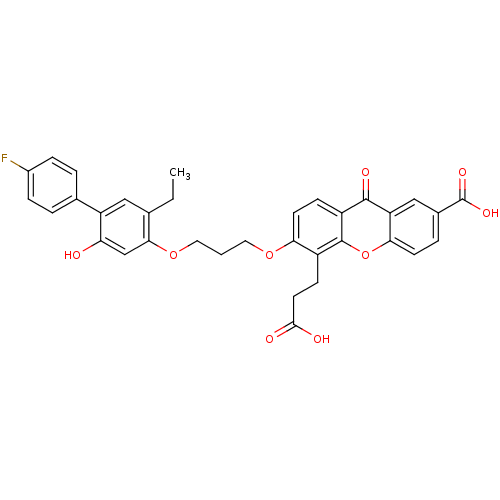
BDBM50029464 5-(2-Carboxy-ethyl)-6-[3-(5-ethyl-4'-fluoro-2-hydroxy-biphenyl-4-yloxy)-propoxy]-9-oxo-9H-xanthene-2-carboxylic acid::CHEMBL292782::LY-292728
SMILES CCc1cc(c(O)cc1OCCCOc1ccc2c(oc3ccc(cc3c2=O)C(O)=O)c1CCC(O)=O)-c1ccc(F)cc1
InChI Key InChIKey=DCTKEJXAVFAMFK-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 7 hits for monomerid = 50029464
Found 7 hits for monomerid = 50029464
Affinity DataKi: 0.0400nMAssay Description:LTB4 receptor antagonist activity was determined by inhibition of specific binding of [3H]-LTB4 in guinea pig lung membranesMore data for this Ligand-Target Pair
Affinity DataKi: 0.0400nMAssay Description:Tested for inhibition of specific binding of [3H]LTB4 to guinea pig lung membranes expressing LTB4 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.470nMAssay Description:LTB4 receptor antagonist activity was determined by inhibition of specific binding of [3H]-LTB4 in human neutrophilMore data for this Ligand-Target Pair
Affinity DataKi: 0.470nMAssay Description:Tested for inhibition of specific binding of [3H]-LTB4 to human neutrophil expressing LTB4 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.470nMAssay Description:Binding affinity at leukotriene B4 receptor on intact human PMNs by displacement of [3H]-LTB4.More data for this Ligand-Target Pair
Affinity DataKi: 0.470nMAssay Description:Compound was tested for inhibitory activity against human neutrophil LTB4 receptor bindingMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMAssay Description:Inhibition of LTB4-induced up-regulation of human neutrophil CD11b/CD18 integrinMore data for this Ligand-Target Pair