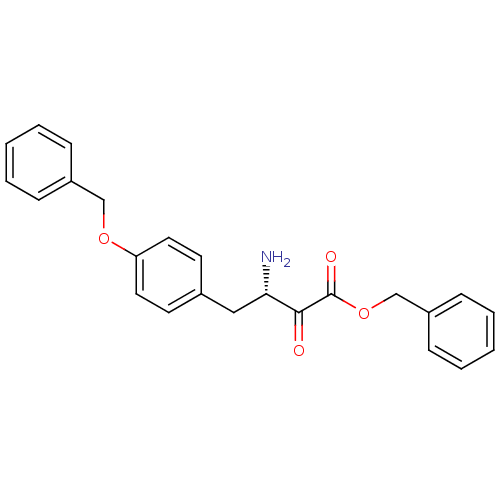
BDBM50046314 3-Amino-4-(4-benzyloxy-phenyl)-2-oxo-butyric acid benzyl ester::CHEMBL71535
SMILES N[C@@H](Cc1ccc(OCc2ccccc2)cc1)C(=O)C(=O)OCc1ccccc1
InChI Key InChIKey=KPMSBJXCMIJUDG-QFIPXVFZSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50046314
Found 4 hits for monomerid = 50046314
Affinity DataKi: 46nMAssay Description:Ability to inhibit amidase activity of LTA4 hydrolase (1.4 ug) purified from human leukocytesMore data for this Ligand-Target Pair
TargetBifunctional epoxide hydrolase 2(Mus musculus (Mouse))
Scripps Research Institute
Curated by ChEMBL
Scripps Research Institute
Curated by ChEMBL
Affinity DataKi: 1.00E+3nMAssay Description:Inhibition constant was determined against epoxide hydrolaseMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory concentration against cytosolic leucine aminopeptidaseMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory concentration against aminopeptidase MMore data for this Ligand-Target Pair