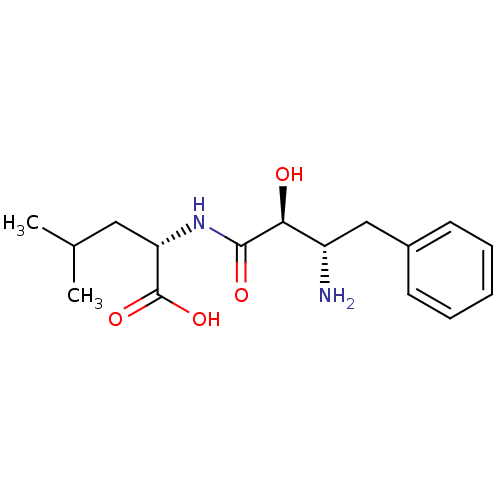
BDBM50046325 (S)-2-((2S,3S)-3-Amino-2-hydroxy-4-phenyl-butyrylamino)-4-methyl-pentanoic acid::2-(3-Amino-2-hydroxy-4-phenyl-butyrylamino)-4-methyl-pentanoic acid::CHEMBL70878
SMILES CC(C)C[C@H](NC(=O)[C@@H](O)[C@@H](N)Cc1ccccc1)C(O)=O
InChI Key InChIKey=VGGGPCQERPFHOB-IHRRRGAJSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50046325
Found 5 hits for monomerid = 50046325
Affinity DataKi: 1.70E+4nMAssay Description:Compound was evaluated for the inhibition of Leucine aminopeptidase and the inhibition constant was determined after preincubating the enzyme and inh...More data for this Ligand-Target Pair
Affinity DataKi: 2.00E+5nMAssay Description:Effect of inhibitor structure on the slow binding inhibition of aminopeptidase M was determined and Ki* was reported which is obtained by the equatio...More data for this Ligand-Target Pair
Affinity DataKi: 5.00E+5nMAssay Description:Effect of inhibitor structure on the slow binding inhibition of aminopeptidase M was determined and the Ki was reported which is = k2/k1More data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+4nMAssay Description:Inhibition of amidase activity of LTA4 hydrolase purified from human leukocytesMore data for this Ligand-Target Pair
Affinity DataIC50: 4.20E+3nMAssay Description:Inhibition of rat liver aminopeptidase B using 0.2 mM L-leucine-beta-naphthylamide by colorimetryMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)