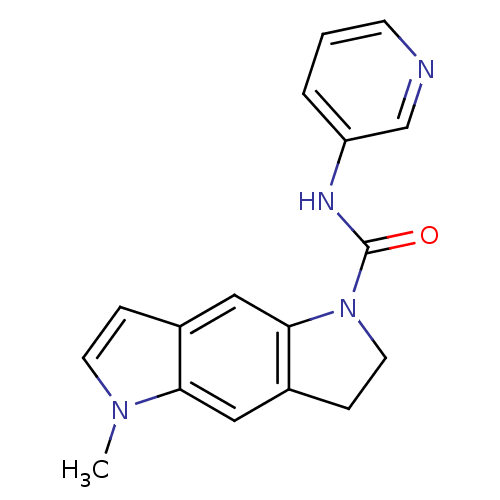
BDBM50060417 5-Methyl-3,5-dihydro-2H-pyrrolo[2,3-f]indole-1-carboxylic acid pyridin-3-ylamide::CHEMBL297784::SB 206553::SB-206553
SMILES Cn1ccc2cc3N(CCc3cc12)C(=O)Nc1cccnc1
InChI Key InChIKey=QJQORSLQNXDVGE-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 38 hits for monomerid = 50060417
Found 38 hits for monomerid = 50060417
Target5-hydroxytryptamine receptor 2B(RAT)
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Affinity DataKi: 2.80nMAssay Description:Displacement of [3H]LSD from human recombinant 5-HT2C receptor expressed in HEK cells by radioligand completion assay relative to controlMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
Institut De Recherches Servier
Curated by PDSP Ki Database
Institut De Recherches Servier
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2B(Homo sapiens (Human))
University Of North Carolina At Chapel Hill
Curated by ChEMBL
University Of North Carolina At Chapel Hill
Curated by ChEMBL
Affinity DataKi: 4.30nMAssay Description:Displacement of [3H]LSD from human cloned 5HT2B receptor expressed in CHO cells by liquid scintillation countingMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Rattus norvegicus (Rat))
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Smithkline Beecham Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 5.01nMAssay Description:Displacement of [3H]mesulergine from rat 5-HT2C receptor expressed in HEK293More data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2B(Homo sapiens (Human))
University Of North Carolina At Chapel Hill
Curated by ChEMBL
University Of North Carolina At Chapel Hill
Curated by ChEMBL
Affinity DataKi: 10nMAssay Description:Binding affinity analysed on 5-HT 2C in human clone using [3H]mesulergine as radioligandMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
Institut De Recherches Servier
Curated by PDSP Ki Database
Institut De Recherches Servier
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
Institut De Recherches Servier
Curated by PDSP Ki Database
Institut De Recherches Servier
Curated by PDSP Ki Database
Affinity DataKi: 13nMAssay Description:Binding affinity towards cloned human 5-hydroxytryptamine 2C receptor expressed in HEK 293 cells using [3H]mesulergine as radioligandMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
Institut De Recherches Servier
Curated by PDSP Ki Database
Institut De Recherches Servier
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2B(Homo sapiens (Human))
University Of North Carolina At Chapel Hill
Curated by ChEMBL
University Of North Carolina At Chapel Hill
Curated by ChEMBL
Affinity DataKi: 20nMAssay Description:Displacement of [3H]LSD from 5-HT2B receptor (unknown origin) assessed as inhibition constant incubated for 90 mins by MicroBeta scintillation counti...More data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2B(Homo sapiens (Human))
University Of North Carolina At Chapel Hill
Curated by ChEMBL
University Of North Carolina At Chapel Hill
Curated by ChEMBL
Target5-hydroxytryptamine receptor 2B(Homo sapiens (Human))
University Of North Carolina At Chapel Hill
Curated by ChEMBL
University Of North Carolina At Chapel Hill
Curated by ChEMBL
Affinity DataKi: 25nMAssay Description:Binding affinity towards cloned human 5-hydroxytryptamine 2B receptor expressed in HEK 293 cells using [3H]5-HT as radioligandMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 7(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 1D(GUINEA PIG)
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Affinity DataKi: 1.62E+3nMAssay Description:Binding affinity analysed on 5-HT 2A human clone using [3H]ketanserin as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 2.00E+3nMAssay Description:Binding affinity towards human 5-hydroxytryptamine 2A receptor expressed in HEK 293 cells using [3H]ketanserin as radioligandMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 1F(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 1E(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
TargetHistamine H1 receptor(Cavia porcellus (domestic guinea pig))
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
TargetGamma-aminobutyric acid receptor subunit alpha-1(Rattus norvegicus (Rat))
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
TargetD(4) dopamine receptor(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
TargetD(3) dopamine receptor(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
TargetAlpha-1A adrenergic receptor(Rattus norvegicus (Rat))
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
TargetD(2) dopamine receptor(Homo sapiens (Human))
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 3A(RAT)
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
TargetAdenosine receptor A1(Rattus norvegicus (rat))
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
TargetGamma-aminobutyric acid receptor subunit alpha-1(Rattus norvegicus (Rat))
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
Smithkline Beecham Pharmaceuticals
Curated by PDSP Ki Database
TargetNeuronal acetylcholine receptor subunit alpha-7(Homo sapiens (Human))
Targacept
Curated by ChEMBL
Targacept
Curated by ChEMBL
Affinity DataEC50: 1.50E+3nMAssay Description:Agonist activity at human alpha7 nAChR expressed in GH4C1 cells assessed as potentiation of nicotine-induced calcium signalMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2B(Homo sapiens (Human))
University Of North Carolina At Chapel Hill
Curated by ChEMBL
University Of North Carolina At Chapel Hill
Curated by ChEMBL
Affinity DataEC50: 13nMAssay Description:Antagonist activity at recombinant human 5-HT2B receptor expressed in CHOK1 cells assessed as inhibition of serotonin-induced increase in inositol1-p...More data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
Institut De Recherches Servier
Curated by PDSP Ki Database
Institut De Recherches Servier
Curated by PDSP Ki Database
Affinity DataIC50: 9.40nMAssay Description:Antagonist activity at human recombinant 5-HT2C receptor assessed as inhibition of serotonin-induced inositol phosphate accumulationMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of heterologously expressed human Cytochrome P450 2C9 at 100 uMMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+4nMAssay Description:Inhibition of heterologously expressed human Cytochrome P450 2D6 at 10 uMMore data for this Ligand-Target Pair
Affinity DataIC50: 50nMAssay Description:Inhibition of heterologously expressed human Cytochrome P450 1A2 at 500 uMMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of heterologously expressed human Cytochrome P450 19A1 at 100 uMMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of heterologously expressed human Cytochrome P450 3A at 1 uMMore data for this Ligand-Target Pair