BDBM50124629 CHEMBL3622879
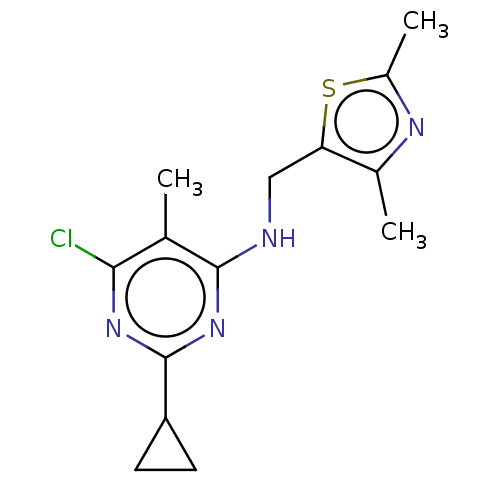
SMILES Cc1nc(C)c(CNc2nc(nc(Cl)c2C)C2CC2)s1
InChI Key InChIKey=BKAQDKUFAULKQY-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50124629
Found 4 hits for monomerid = 50124629
TargetcAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataKi: 4.80nMAssay Description:Inhibition of human PDE10A2 transfected in AD293 cells by IMAP FP assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.70E+4nMAssay Description:Inhibition of CYP3A4 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 8.20E+3nMAssay Description:Inhibition of CYP2D6 (unknown origin)More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 1 group I member 2(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 1.90E+4nMAssay Description:Activation of PXR (unknown origin)More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)