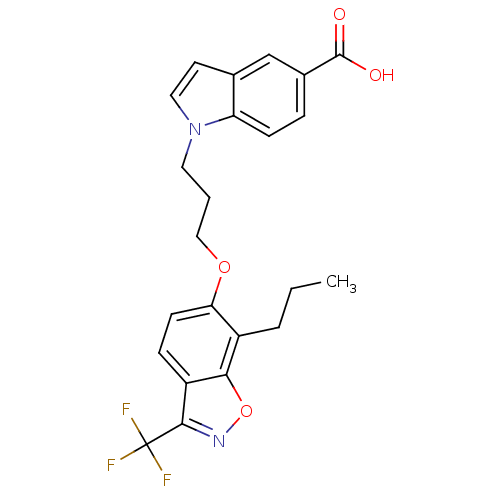
BDBM50184268 1-(3-(7-propyl-3-(trifluoromethyl)benzo[d]isoxazol-6-yloxy)propyl)-1H-indole-5-carboxylic acid::CHEMBL377218
SMILES CCCc1c(OCCCn2ccc3cc(ccc23)C(O)=O)ccc2c(noc12)C(F)(F)F
InChI Key InChIKey=WJPMVXGXXLNBEN-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50184268
Found 5 hits for monomerid = 50184268
TargetOxysterols receptor LXR-beta(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 199nMAssay Description:Inhibition of LXR betaMore data for this Ligand-Target Pair
TargetOxysterols receptor LXR-beta(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 2.80E+3nMAssay Description:Activity at LXR beta as beta-lactamase transactivation in CHO cellsMore data for this Ligand-Target Pair
TargetOxysterols receptor LXR-alpha(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 72nMAssay Description:Activity at LXR alpha as beta-lactamase transactivation in CHO cellsMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor alpha(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 8.30E+3nMAssay Description:Inhibition of PPAR alphaMore data for this Ligand-Target Pair
TargetOxysterols receptor LXR-alpha(Homo sapiens (Human))
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 1.90E+3nMAssay Description:Activity at LXR alpha as beta-lactamase transactivation in CHO cellsMore data for this Ligand-Target Pair