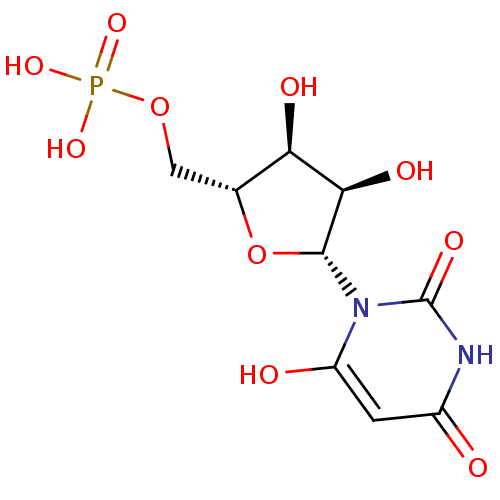
BDBM50199178 1-beta-D-ribofuranosyl(3H)pyrimidine-2,4,6-trione 5'-monophosphate::3,4-dihydroxy-5-(6-oxido-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)tetrahydrofuran-2-yl)methyl phosphate::6-HYDROXYURIDINE-5'-PHOSPHATE::6-hydroxy-UMP::CHEMBL383923
SMILES O[C@@H]1[C@@H](COP(O)(O)=O)O[C@H]([C@@H]1O)n1c(O)cc(=O)[nH]c1=O
InChI Key InChIKey=UDOBICLZEKUKCV-YXZULKJRSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50199178
Found 5 hits for monomerid = 50199178
TargetOrotidine 5'-phosphate decarboxylase(Saccharomyces cerevisiae)
University Health Network
Curated by ChEMBL
University Health Network
Curated by ChEMBL
Affinity DataKi: 0.00880nMAssay Description:Inhibition of Saccharomyces cerevisia uridine 5'-monophosphate synthaseMore data for this Ligand-Target Pair
TargetOrotidine 5'-phosphate decarboxylase(Saccharomyces cerevisiae)
University Health Network
Curated by ChEMBL
University Health Network
Curated by ChEMBL
Affinity DataKi: 0.00900nM ΔG°: -15.1kcal/moleT: 2°CAssay Description:Inhibition of Saccharomyces cerevisia uridine 5'-monophosphate synthase after overnight incubation at room temperature by VP-ITC microcalorimetryMore data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+5nMAssay Description:Agonist activity at human P2Y6 receptor expressed in 1321N1 cells assessed as IP accumulation by SPAMore data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+5nMAssay Description:Agonist activity at human P2Y4 receptor expressed in 1321N1 cells assessed as IP accumulation by SPAMore data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+5nMAssay Description:Agonist activity at human P2Y2 receptor expressed in 1321N1 cells assessed as IP accumulation by SPAMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)