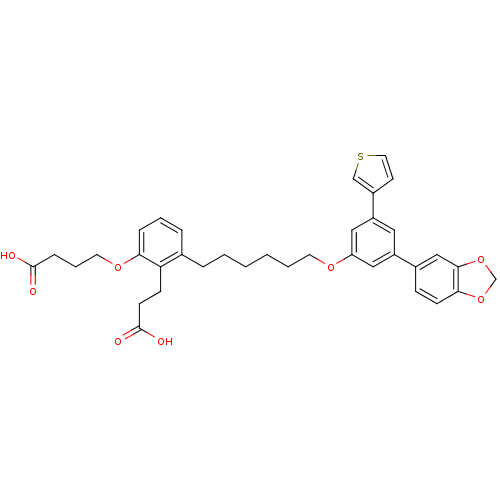
BDBM50317626 4-{3-[6-(3-5-Benzo[1,3]dioxolyl-5-thiophen-3-ylphenoxy)hexyl]-2-(2-carboxyethyl)phenoxy}butyric Acid::CHEMBL1098560
SMILES OC(=O)CCCOc1cccc(CCCCCCOc2cc(cc(c2)-c2ccc3OCOc3c2)-c2ccsc2)c1CCC(O)=O
InChI Key InChIKey=SRPFDFNVILUDAI-UHFFFAOYSA-N
Data 10 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 10 hits for monomerid = 50317626
Found 10 hits for monomerid = 50317626
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Roche Research Center
Curated by ChEMBL
Roche Research Center
Curated by ChEMBL
Affinity DataIC50: >3.00E+3nMAssay Description:Inhibition of human ERG expressed in CHO cells assessed as reduction of current amplitudeMore data for this Ligand-Target Pair
Affinity DataIC50: 114nMAssay Description:Antagonist activity at FLAG-tagged human BLT1 receptor expressed in HEK293 cells assessed as inhibition of LTB4-stimulated calcium mobilization prein...More data for this Ligand-Target Pair
Affinity DataIC50: 164nMAssay Description:Antagonist activity at FLAG-tagged human BLT2 receptor expressed in HEK293 cells assessed as inhibition of LTB4-stimulated calcium mobilization prein...More data for this Ligand-Target Pair
Affinity DataIC50: >7.50E+3nMAssay Description:Inhibition of CYP1A2 by fluorescence methodMore data for this Ligand-Target Pair
Affinity DataIC50: 3.90E+3nMAssay Description:Inhibition of CYP2C9 fluorescence methodMore data for this Ligand-Target Pair
Affinity DataIC50: >7.50E+3nMAssay Description:Inhibition of CYP2C19 flourescence methodMore data for this Ligand-Target Pair
Affinity DataIC50: >7.50E+3nMAssay Description:Inhibition of CYP3A4 fluorescence methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.440nMAssay Description:Antagonist activity at BLT1 receptor expressed in human HL60 cells assessed as inhibition of LTB4-stimulated calcium flux after 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.25E+4nMAssay Description:Inhibition of CYP2C9 by LC/MS methodMore data for this Ligand-Target Pair
Affinity DataIC50: >7.50E+3nMAssay Description:Inhibition of CYP2D6 flourescence methodMore data for this Ligand-Target Pair