BDBM50383378 CHEMBL2030561
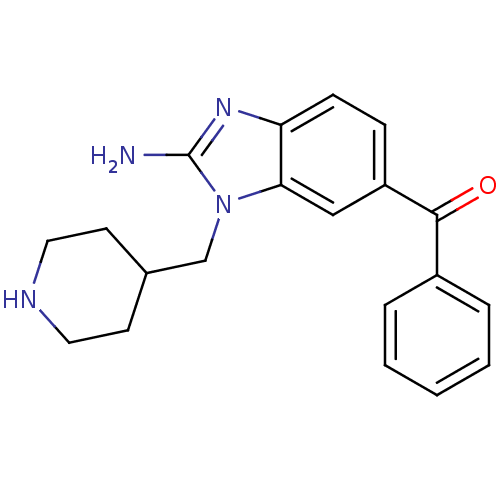
SMILES Nc1nc2ccc(cc2n1CC1CCNCC1)C(=O)c1ccccc1
InChI Key InChIKey=CMCWMODUNMLBRJ-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50383378
Found 5 hits for monomerid = 50383378
TargetProto-oncogene tyrosine-protein kinase Src(Homo sapiens (Human))
University Of Washington
Curated by ChEMBL
University Of Washington
Curated by ChEMBL
Affinity DataKi: >1.00E+4nMAssay Description:Inhibition of human SRC by radiometric assayMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:Inhibition of human ABL by radiometric assayMore data for this Ligand-Target Pair
TargetProto-oncogene tyrosine-protein kinase Src(Homo sapiens (Human))
University Of Washington
Curated by ChEMBL
University Of Washington
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human SRCMore data for this Ligand-Target Pair
TargetCalmodulin-domain protein kinase 1(Toxoplasma gondii)
University Of Washington
Curated by ChEMBL
University Of Washington
Curated by ChEMBL
Affinity DataIC50: 15nMAssay Description:Inhibition of Toxoplasma gondii CDPK1More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of human ABLMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)