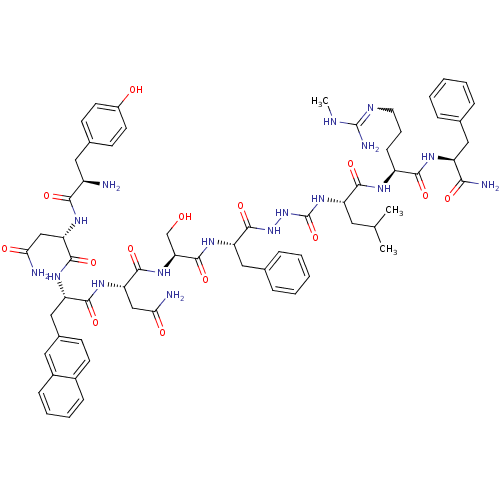
BDBM50392412 CHEMBL2151653
SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)NNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1ccc2ccccc2c1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O
InChI Key InChIKey=VZQWRKAHIZUTDA-JMQQQMJGSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50392412
Found 3 hits for monomerid = 50392412
Affinity DataEC50: 0.0980nMAssay Description:Agonist activity at human KISS1R assessed as induction of intracellular calcium mobilization by fluorometric analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Binding affinity to rat KISS1RMore data for this Ligand-Target Pair
Affinity DataIC50: 0.130nMAssay Description:Binding affinity to human KISS1RMore data for this Ligand-Target Pair