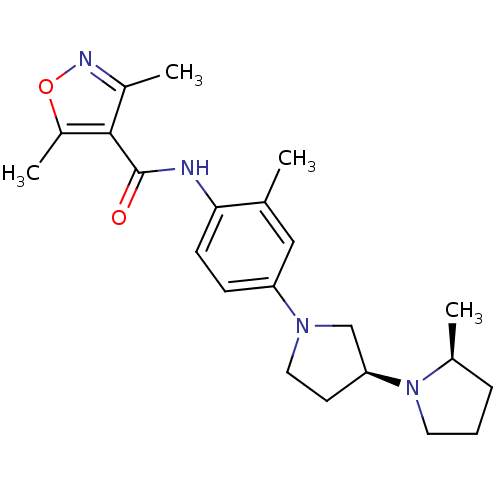
BDBM50443217 CHEMBL3087669
SMILES C[C@H]1CCCN1[C@H]1CCN(C1)c1ccc(NC(=O)c2c(C)noc2C)c(C)c1
InChI Key InChIKey=CJUSJFAZUSWFLY-KXBFYZLASA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50443217
Found 5 hits for monomerid = 50443217
Affinity DataKi: 0.400nMAssay Description:Displacement of [3H]N-alpha-methylhistamine from recombinant rhesus monkey histamine H3 receptor transfected in CHO cells after 1 hr by scintillation...More data for this Ligand-Target Pair
Affinity DataKi: 0.900nMAssay Description:Displacement of [3H]-methylhistamine from rat histamine H3 receptor (445 amino acid residues) transfected in human 293 cells after 1 hr by scintillat...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of CYP2C9 (unknown origin) after 4 hrsMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Canis familiaris)
Sanofi Us
Curated by ChEMBL
Sanofi Us
Curated by ChEMBL
Affinity DataIC50: 4.40E+4nMAssay Description:Inhibition of canine ERG current transfected in CHO cells by patch clamp assayMore data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of CYP2C19 (unknown origin) after 4 hrsMore data for this Ligand-Target Pair