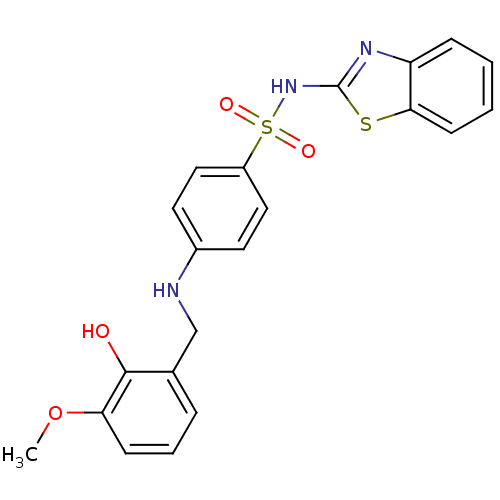
BDBM50447175 CHEMBL3113165::US10752581, Compound 35
SMILES COc1cccc(CNc2ccc(cc2)S(=O)(=O)Nc2nc3ccccc3s2)c1O
InChI Key InChIKey=OWHBVKBNNRYMIN-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 10 hits for monomerid = 50447175
Found 10 hits for monomerid = 50447175
TargetPolyunsaturated fatty acid lipoxygenase ALOX12(Homo sapiens (Human))
National Institutes Of Health
Curated by ChEMBL
National Institutes Of Health
Curated by ChEMBL
Affinity DataKi: 350nMAssay Description:Uncompetitive inhibition of N-terminal His6-tagged human platelet 12-LOX using arachidonic acid as substrate assessed as 12-HPETE formation by Henri-...More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid lipoxygenase ALOX12(Homo sapiens (Human))
National Institutes Of Health
Curated by ChEMBL
National Institutes Of Health
Curated by ChEMBL
Affinity DataKi: 720nMAssay Description:Competitive inhibition of N-terminal His6-tagged human platelet 12-LOX using arachidonic acid as substrate assessed as 12-HPETE formation by Henri-Mi...More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid lipoxygenase ALOX12(Homo sapiens (Human))
National Institutes Of Health
Curated by ChEMBL
National Institutes Of Health
Curated by ChEMBL
Affinity DataIC50: 360nMAssay Description:Inhibition of wild type human N-terminal His-tagged 12-LOX using arachidonic acid as substrate by UV-Vis spectroscopy analysisMore data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid lipoxygenase ALOX15(Homo sapiens (Human))
National Institutes Of Health
Curated by ChEMBL
National Institutes Of Health
Curated by ChEMBL
Affinity DataIC50: 9.70E+3nMAssay Description:Inhibition of N-terminal His6-tagged human reticulocyte 15-LOX1 using arachidonic acid as substrate by UV-vis spectrophotometric analysisMore data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid lipoxygenase ALOX12(Homo sapiens (Human))
National Institutes Of Health
Curated by ChEMBL
National Institutes Of Health
Curated by ChEMBL
Affinity DataIC50: 340nMAssay Description:Inhibition of N-terminal His6-tagged human platelet 12-LOX using arachidonic acid as substrate by UV-vis spectrophotometric analysisMore data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid lipoxygenase ALOX12(Homo sapiens (Human))
National Institutes Of Health
Curated by ChEMBL
National Institutes Of Health
Curated by ChEMBL
Affinity DataIC50: 340nMAssay Description:UV-vis cuvette-based assay.More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid lipoxygenase ALOX15(Homo sapiens (Human))
National Institutes Of Health
Curated by ChEMBL
National Institutes Of Health
Curated by ChEMBL
Affinity DataIC50: 9.70E+3nMAssay Description:UV-vis cuvette-based assay.More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
National Institutes Of Health
Curated by ChEMBL
National Institutes Of Health
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of human 5-LOX using arachidonic acid as substrate by UV-vis spectrophotometric analysisMore data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid lipoxygenase ALOX12(Mus musculus)
National Institutes Of Health
Curated by ChEMBL
National Institutes Of Health
Curated by ChEMBL
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of 12-LOX in mouse BTC3 cells assessed as inhibition of arachidonic acid/calcium ionophore-induced 12-HETE production after 4 hrs by ELISAMore data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid lipoxygenase ALOX15B(Homo sapiens (Human))
National Institutes Of Health
Curated by ChEMBL
National Institutes Of Health
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of N-terminal His6-tagged human epithelial 15-LOX2 using arachidonic acid as substrate by UV-vis spectrophotometric analysisMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)