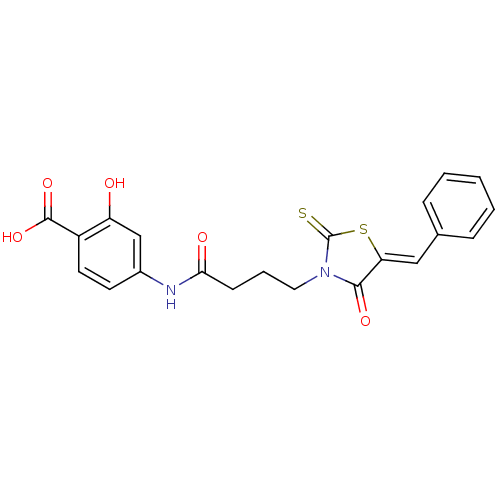
BDBM61652 2-hydroxy-4-[[1-oxo-4-[(5Z)-4-oxo-5-(phenylmethylene)-2-sulfanylidene-3-thiazolidinyl]butyl]amino]benzoic acid::2-oxidanyl-4-[4-[(5Z)-4-oxidanylidene-5-(phenylmethylidene)-2-sulfanylidene-1,3-thiazolidin-3-yl]butanoylamino]benzoic acid::4-[4-[(5Z)-5-benzal-4-keto-2-thioxo-thiazolidin-3-yl]butanoylamino]-2-hydroxy-benzoic acid::4-[4-[(5Z)-5-benzylidene-4-oxo-2-sulfanylidene-1,3-thiazolidin-3-yl]butanoylamino]-2-hydroxybenzoic acid::MLS001157852::SMR000651252::cid_6226024
SMILES OC(=O)c1ccc(NC(=O)CCCN2C(=S)S\C(=C/c3ccccc3)C2=O)cc1O
InChI Key InChIKey=OSZBSJXLNJYIRY-BOPFTXTBSA-N
Data 7 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 7 hits for monomerid = 61652
Found 7 hits for monomerid = 61652
TargetG-protein coupled receptor 35(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 500nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetG-protein coupled receptor 55(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: >3.20E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics(SSBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetNucleotide-binding oligomerization domain-containing protein 1(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 295nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics Source Affiliation: Sanford-Burnham Medical Research Institute Network: NIH Molecular Lib...More data for this Ligand-Target Pair
TargetDNA (cytosine-5)-methyltransferase 3A(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 1.72E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 22(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: >1.00E+5nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 22(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 8.50E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, CA...More data for this Ligand-Target Pair
TargetG-protein coupled receptor 35(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 2.67E+3nMAssay Description:Data Source: Dr. Mary Abood Source Affiliation: Temple University Network: NIH Molecular Libraries Probe Production Centers Network (MLPCN) Grant Pro...More data for this Ligand-Target Pair