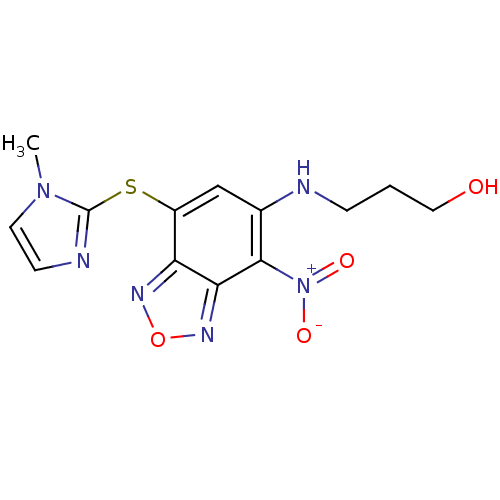
BDBM63626 3-({7-[(1-methyl-1H-imidazol-2-yl)thio]-4-nitro-2,1,3-benzoxadiazol-5-yl}amino)propan-1-ol::3-[[7-(1-methylimidazol-2-yl)sulfanyl-4-nitro-2,1,3-benzoxadiazol-5-yl]amino]propan-1-ol::3-[[7-[(1-methyl-2-imidazolyl)thio]-4-nitro-2,1,3-benzoxadiazol-5-yl]amino]-1-propanol::3-[[7-[(1-methylimidazol-2-yl)thio]-4-nitro-benzofurazan-5-yl]amino]propan-1-ol::MLS000674026::SMR000311304::cid_4326878
SMILES Cn1ccnc1Sc1cc(NCCCO)c([N+]([O-])=O)c2nonc12
InChI Key InChIKey=MOEPPVLXSWWAAN-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 63626
Found 4 hits for monomerid = 63626
Affinity DataEC50: 1.18E+4nMAssay Description:Keywords: Heat Shock Factor-1 (HSF-1), Stress Response, MG132, NIH3T3, Luminescence Assay Overview: Modified NIH3T3, transformed to express firefly...More data for this Ligand-Target Pair
TargetReceptor-interacting serine/threonine-protein kinase 2(Homo sapiens (Human))
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
TargetBeta-lactamase(Pseudomonas aeruginosa)
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
TargetNeurotensin receptor type 1(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataEC50: 1.32E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair