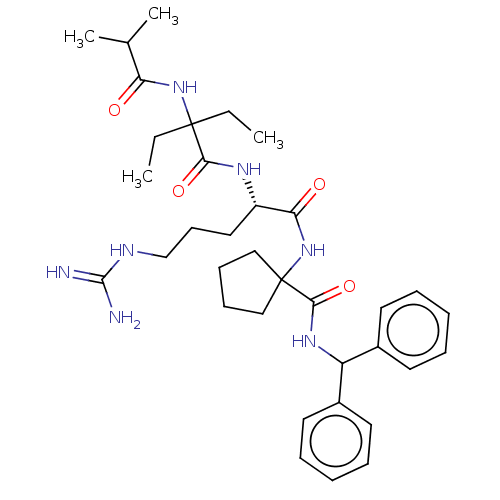
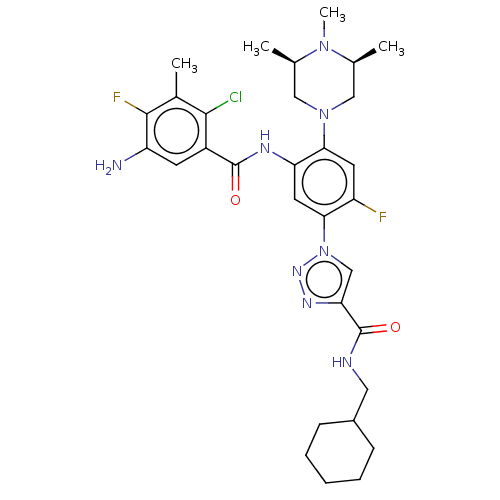
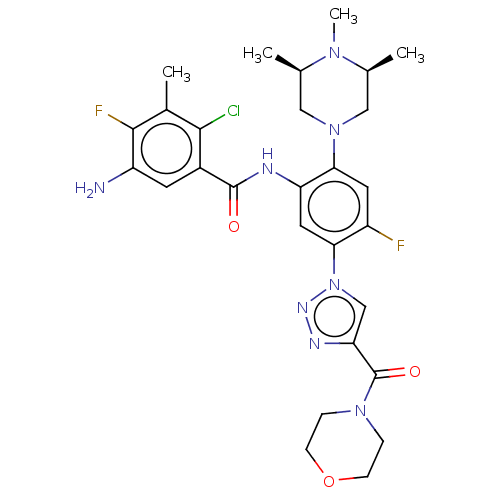
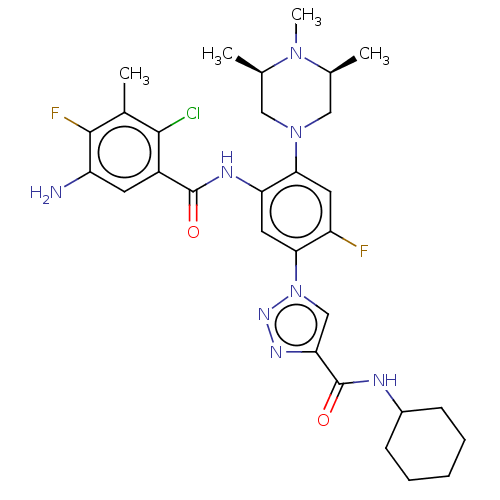
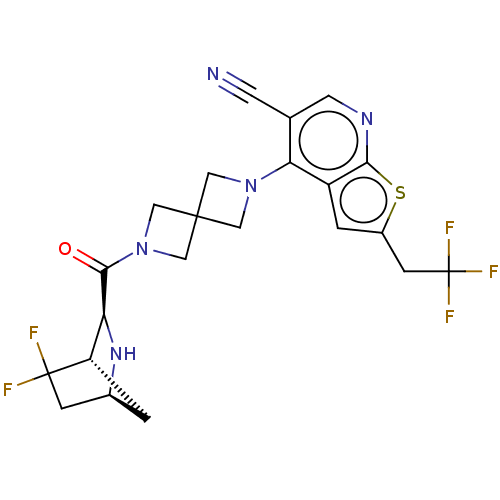
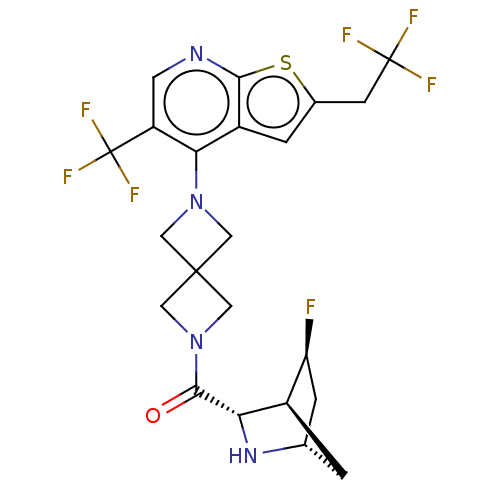
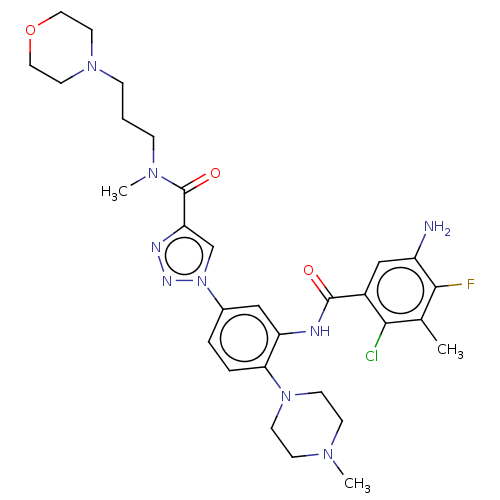
TargetHistone-lysine N-methyltransferase 2A(Homo sapiens (Human))
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataKi: <1nMAssay Description:Inhibition of MLL1 binding to N-terminal His-tagged WRD5 23 deletion mutant (24 to 334 residues) (unknown origin) expressed in Escherichia coli Roset...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A(Homo sapiens (Human))
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataKi: <1nMAssay Description:Inhibition of MLL1 binding to N-terminal His-tagged WRD5 23 deletion mutant (24 to 334 residues) (unknown origin) expressed in Escherichia coli Roset...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A(Homo sapiens (Human))
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataKi: <1nMAssay Description:Inhibition of MLL1 binding to N-terminal His-tagged WRD5 23 deletion mutant (24 to 334 residues) (unknown origin) expressed in Escherichia coli Roset...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A(Homo sapiens (Human))
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 2.40nMAssay Description:Inhibition of MLL1 binding to N-terminal His-tagged WRD5 23 deletion mutant (24 to 334 residues) (unknown origin) expressed in Escherichia coli Roset...More data for this Ligand-Target Pair
Affinity DataIC50: 2.60nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A(Homo sapiens (Human))
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 2.90nMAssay Description:Inhibition of MLL1 binding to N-terminal His-tagged WRD5 23 deletion mutant (24 to 334 residues) (unknown origin) expressed in Escherichia coli Roset...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: <3nMAssay Description:Menin1-615 wherein 6�His tag and HA tag are inserted in the N-terminus, and myc tag is inserted in the C-terminus (hereinafter, referred to as His-Me...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: <3nMAssay Description:Menin1-615 wherein 6�His tag and HA tag are inserted in the N-terminus, and myc tag is inserted in the C-terminus (hereinafter, referred to as His-Me...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: <3nMAssay Description:Menin1-615 wherein 6�His tag and HA tag are inserted in the N-terminus, and myc tag is inserted in the C-terminus (hereinafter, referred to as His-Me...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: <3nMAssay Description:Menin1-615 wherein 6�His tag and HA tag are inserted in the N-terminus, and myc tag is inserted in the C-terminus (hereinafter, referred to as His-Me...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 3.08nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 3.65nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 3.86nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 3.99nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: 4.10nMAssay Description:Menin1-615 wherein 6�His tag and HA tag are inserted in the N-terminus, and myc tag is inserted in the C-terminus (hereinafter, referred to as His-Me...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A(Homo sapiens (Human))
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 4.5nMAssay Description:Inhibition of MLL1 binding to N-terminal His-tagged WRD5 23 deletion mutant (24 to 334 residues) (unknown origin) expressed in Escherichia coli Roset...More data for this Ligand-Target Pair
Affinity DataIC50: 4.72nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 4.91nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 5.24nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: 5.30nMAssay Description:Menin1-615 wherein 6�His tag and HA tag are inserted in the N-terminus, and myc tag is inserted in the C-terminus (hereinafter, referred to as His-Me...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: 6.20nMAssay Description:Menin1-615 wherein 6�His tag and HA tag are inserted in the N-terminus, and myc tag is inserted in the C-terminus (hereinafter, referred to as His-Me...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: 6.20nMAssay Description:Test for Evaluating the Inbinition of the Menin-MLL BindingMenin1-615 wherein 6� His tag and HA tag are inserted in the N-terminus, and myc tag is in...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: 7nMAssay Description:Menin1-615 wherein 6�His tag and HA tag are inserted in the N-terminus, and myc tag is inserted in the C-terminus (hereinafter, referred to as His-Me...More data for this Ligand-Target Pair
Affinity DataIC50: 7.09nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: 7.10nMAssay Description:Menin1-615 wherein 6�His tag and HA tag are inserted in the N-terminus, and myc tag is inserted in the C-terminus (hereinafter, referred to as His-Me...More data for this Ligand-Target Pair
Affinity DataIC50: 7.23nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: 7.90nMAssay Description:Menin1-615 wherein 6�His tag and HA tag are inserted in the N-terminus, and myc tag is inserted in the C-terminus (hereinafter, referred to as His-Me...More data for this Ligand-Target Pair
Affinity DataIC50: 8.23nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: 8.40nMAssay Description:Test for Evaluating the Inbinition of the Menin-MLL BindingMenin1-615 wherein 6� His tag and HA tag are inserted in the N-terminus, and myc tag is in...More data for this Ligand-Target Pair
Affinity DataIC50: 9.12nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 9.62nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 10.8nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: 11nMAssay Description:Test for Evaluating the Inbinition of the Menin-MLL BindingMenin1-615 wherein 6� His tag and HA tag are inserted in the N-terminus, and myc tag is in...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: 11.4nMAssay Description:Test for Evaluating the Inbinition of the Menin-MLL BindingMenin1-615 wherein 6� His tag and HA tag are inserted in the N-terminus, and myc tag is in...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: 13.3nMAssay Description:Test for Evaluating the Inbinition of the Menin-MLL BindingMenin1-615 wherein 6� His tag and HA tag are inserted in the N-terminus, and myc tag is in...More data for this Ligand-Target Pair
Affinity DataIC50: 15.6nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 16.6nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 16.8nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 17.1nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: 17.8nMAssay Description:Test for Evaluating the Inbinition of the Menin-MLL BindingMenin1-615 wherein 6� His tag and HA tag are inserted in the N-terminus, and myc tag is in...More data for this Ligand-Target Pair
Affinity DataIC50: 19.4nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 19.4nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 19.8nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A(Homo sapiens (Human))
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 20nMAssay Description:Inhibition of MLL1 (unknown origin) histone methyltransferase activityMore data for this Ligand-Target Pair
Affinity DataIC50: 20.4nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 20.6nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
Affinity DataIC50: 21.2nMAssay Description:WDR5 TR-FRET Assay Procedure: Stock compounds were transferred to the assay plate by Echo Liquid Handler. Reactions were performed in the assay buffe...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: 26nMAssay Description:Test for Evaluating the Inbinition of the Menin-MLL BindingMenin1-615 wherein 6� His tag and HA tag are inserted in the N-terminus, and myc tag is in...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A/Menin(Homo sapiens (Human))
Sumitomo Dainippon Pharma
US Patent
Sumitomo Dainippon Pharma
US Patent
Affinity DataIC50: 27.3nMAssay Description:Test for Evaluating the Inbinition of the Menin-MLL BindingMenin1-615 wherein 6� His tag and HA tag are inserted in the N-terminus, and myc tag is in...More data for this Ligand-Target Pair