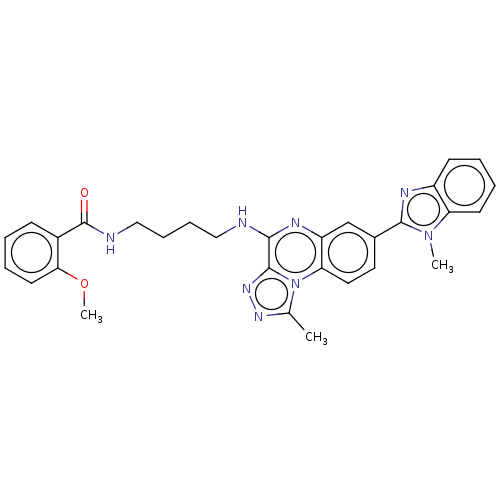
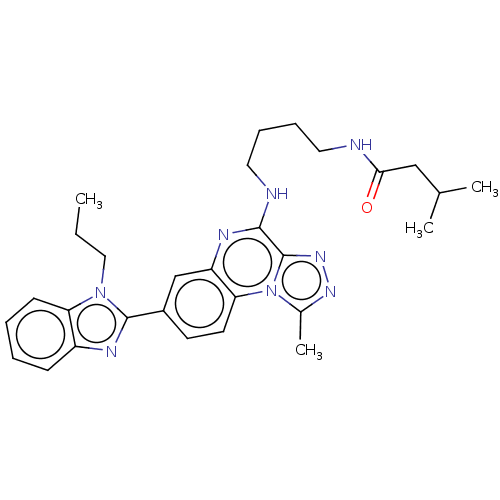
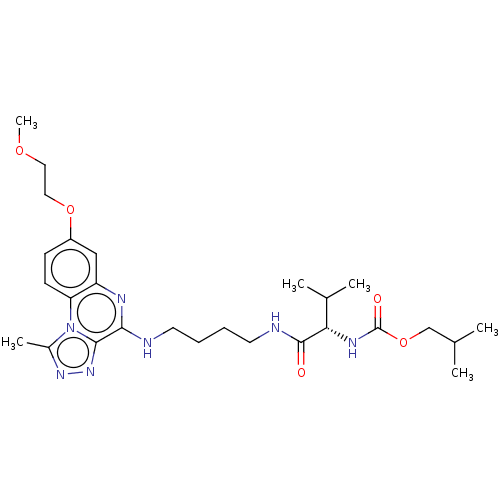
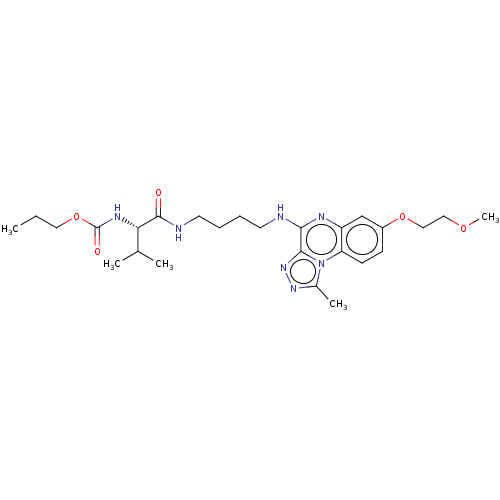
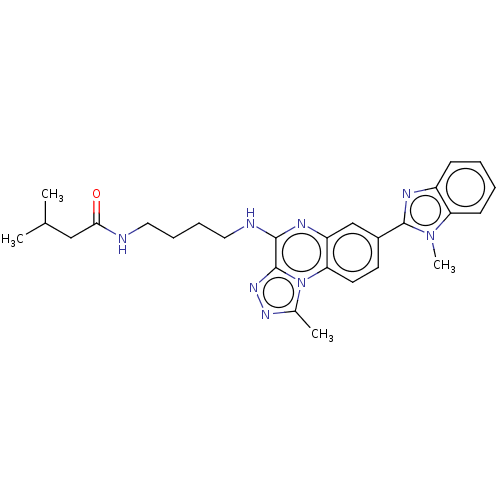
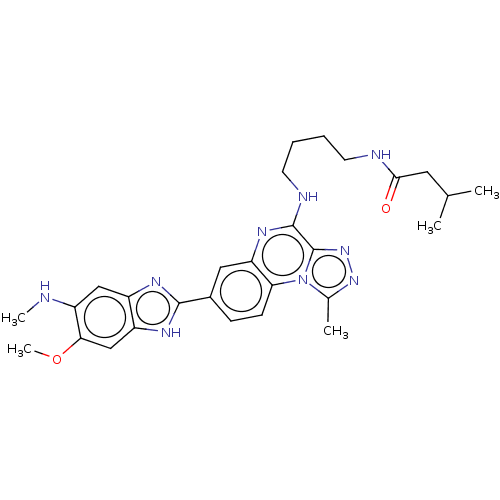
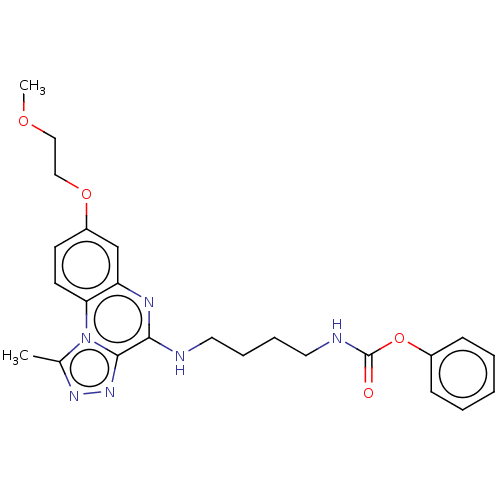
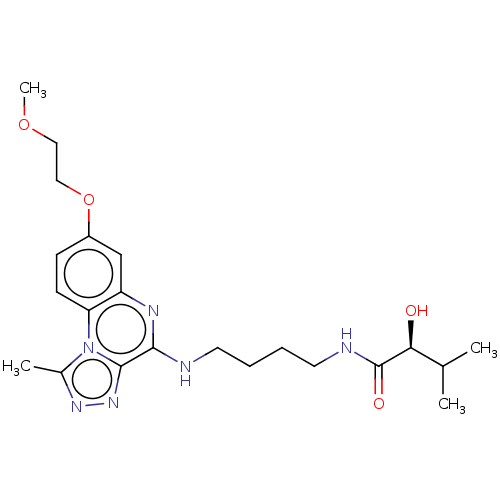
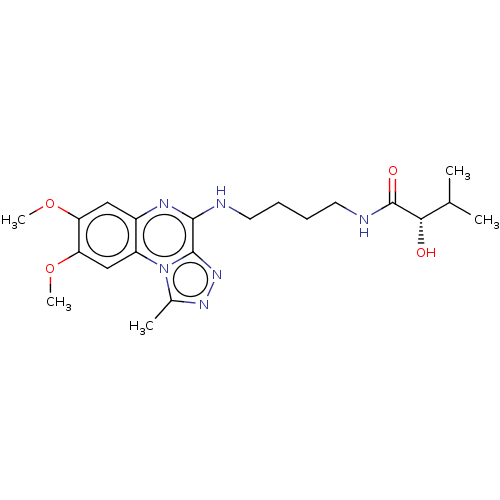
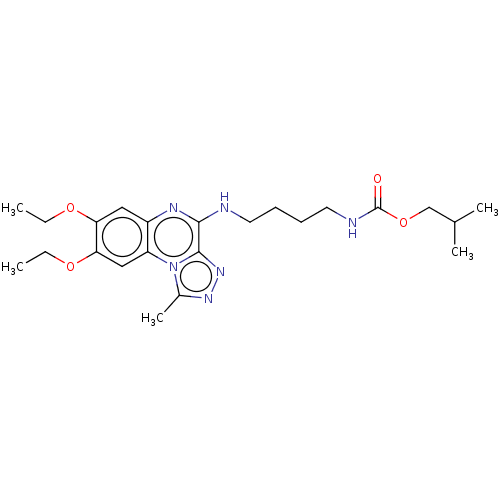
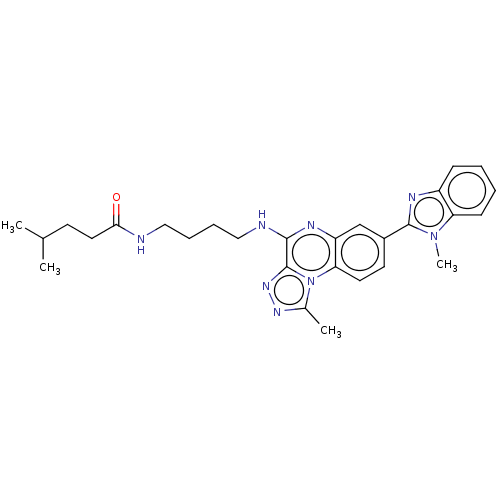
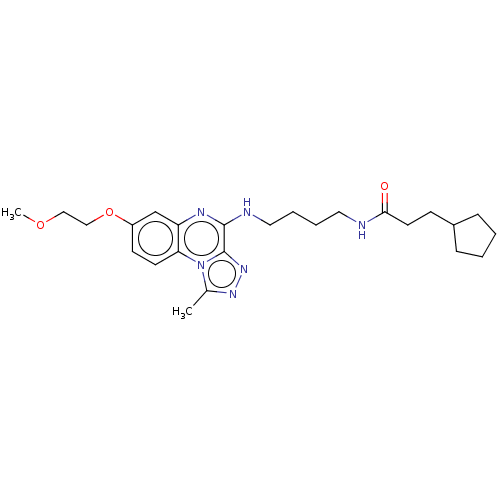
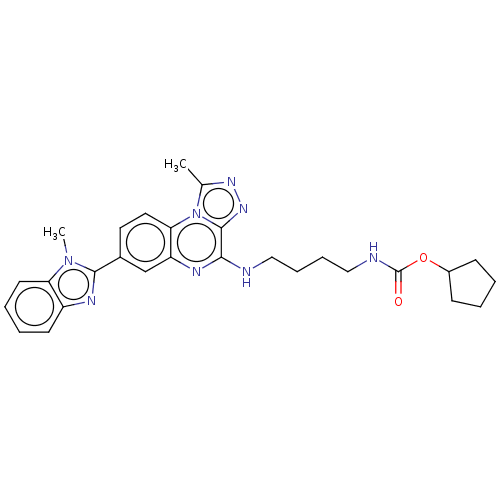
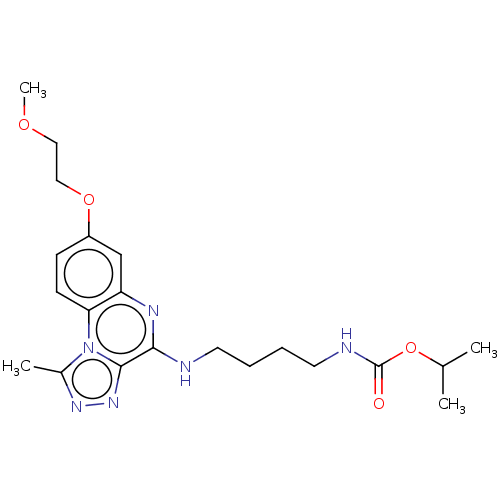
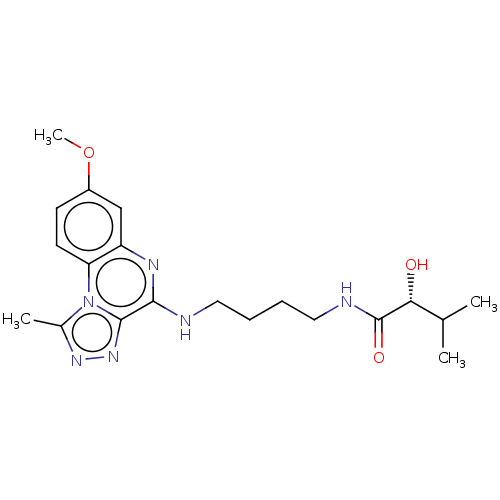
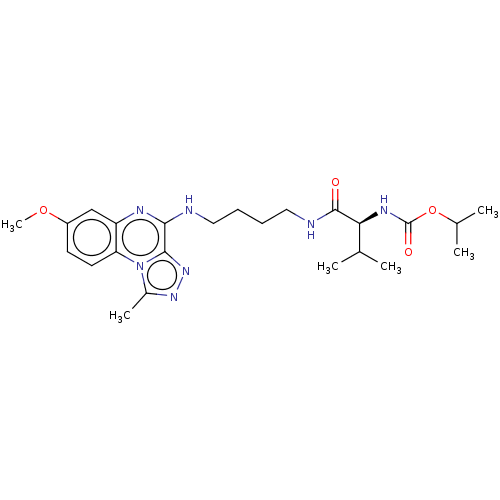
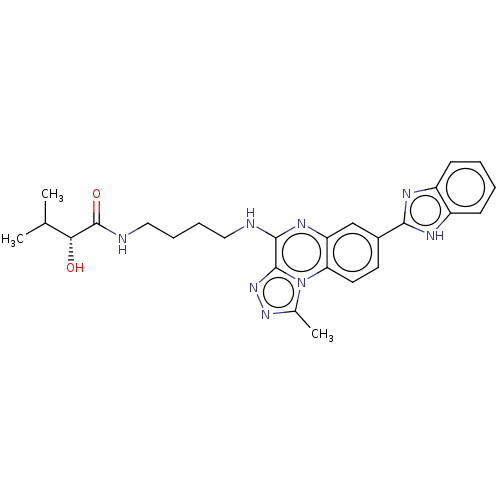
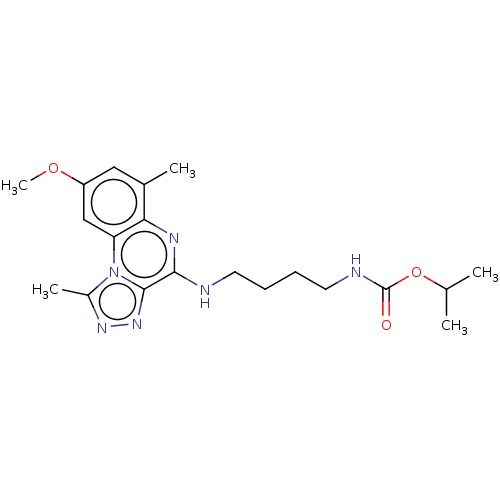
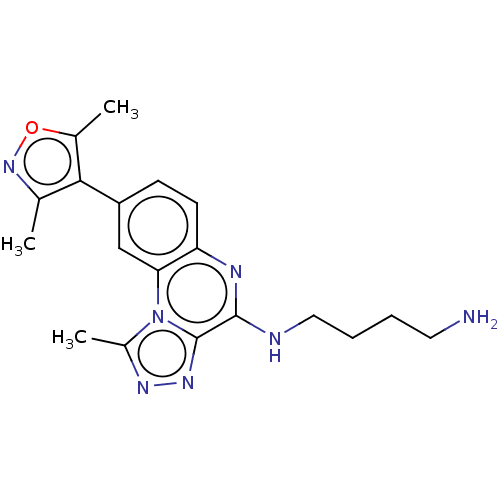
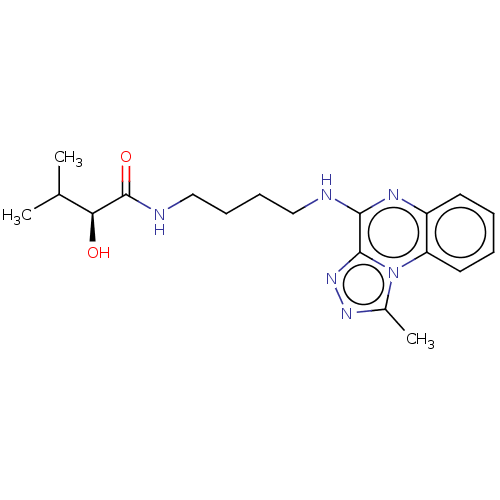
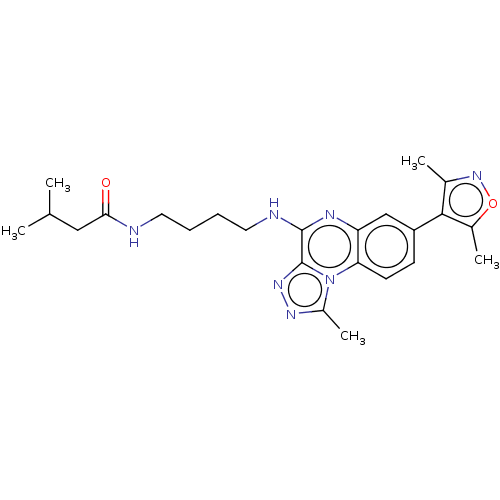
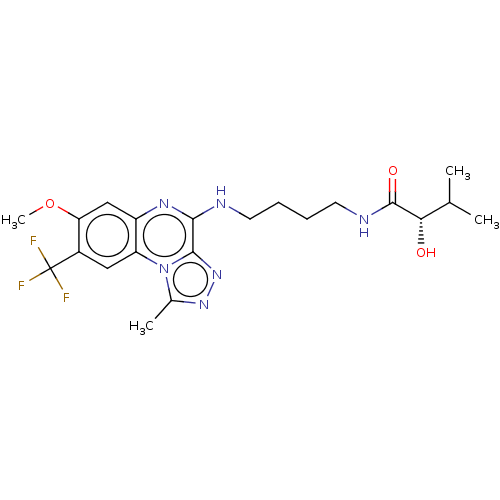
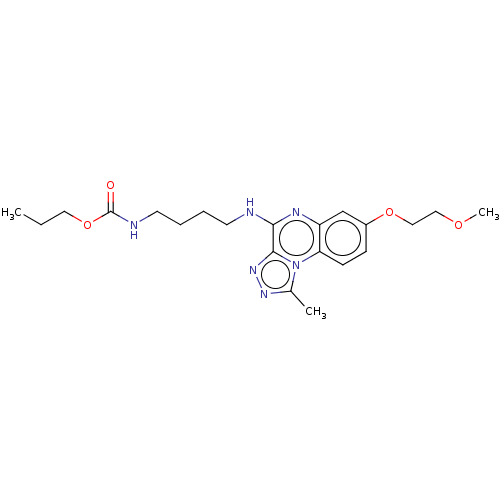
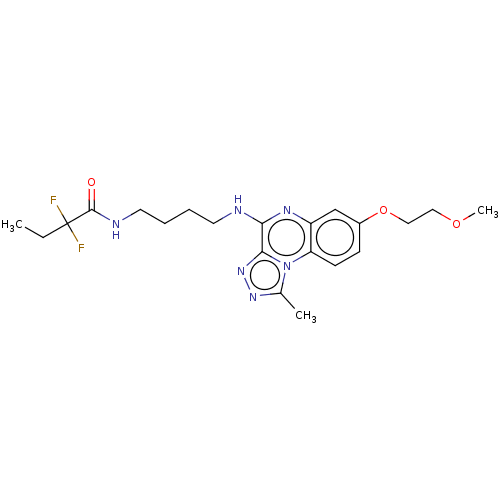
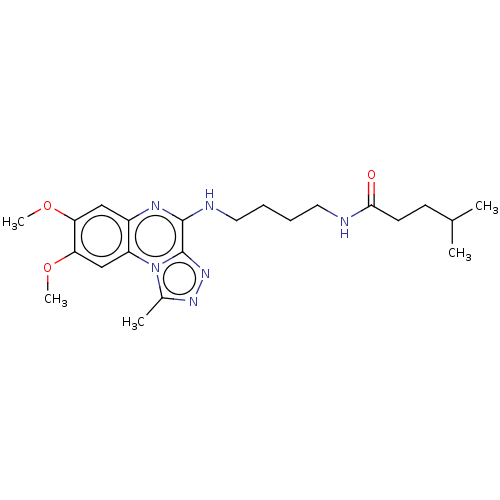
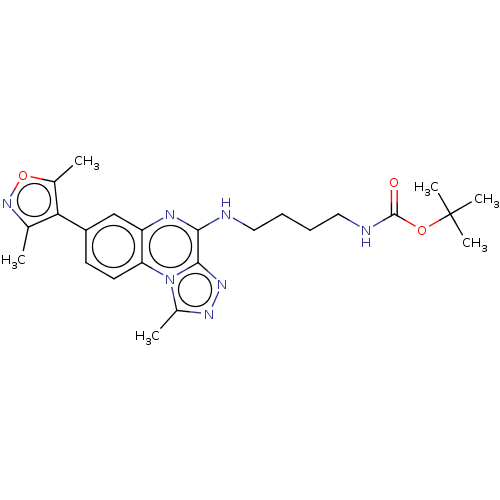
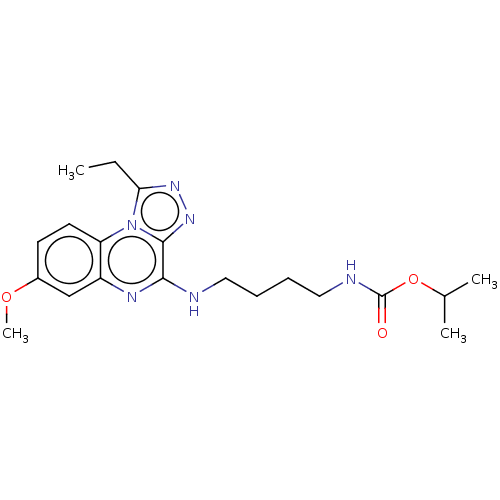
Affinity DataIC50: 0.400nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 0.900nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
TargetCholesteryl ester transfer protein(Homo sapiens (Human))
Chong Kun Dang Pharmaceutical
US Patent
Chong Kun Dang Pharmaceutical
US Patent
Affinity DataIC50: 1.5nMAssay Description:As a protein source for cholesteryl ester transfer, plasma from healthy persons was used, and as a cholesteryl ester receptor, LDL from healthy perso...More data for this Ligand-Target Pair
TargetCholesteryl ester transfer protein(Homo sapiens (Human))
Chong Kun Dang Pharmaceutical
US Patent
Chong Kun Dang Pharmaceutical
US Patent
Affinity DataIC50: 1.60nMAssay Description:As a protein source for cholesteryl ester transfer, plasma from healthy persons was used, and as a cholesteryl ester receptor, LDL from healthy perso...More data for this Ligand-Target Pair
TargetCholesteryl ester transfer protein(Homo sapiens (Human))
Chong Kun Dang Pharmaceutical
US Patent
Chong Kun Dang Pharmaceutical
US Patent
Affinity DataIC50: 1.90nMAssay Description:As a protein source for cholesteryl ester transfer, plasma from healthy persons was used, and as a cholesteryl ester receptor, LDL from healthy perso...More data for this Ligand-Target Pair
TargetCholesteryl ester transfer protein(Homo sapiens (Human))
Chong Kun Dang Pharmaceutical
US Patent
Chong Kun Dang Pharmaceutical
US Patent
Affinity DataIC50: 1.90nMAssay Description:As a protein source for cholesteryl ester transfer, plasma from healthy persons was used, and as a cholesteryl ester receptor, LDL from healthy perso...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
TargetCholesteryl ester transfer protein(Homo sapiens (Human))
Chong Kun Dang Pharmaceutical
US Patent
Chong Kun Dang Pharmaceutical
US Patent
Affinity DataIC50: 2.5nMAssay Description:As a protein source for cholesteryl ester transfer, plasma from healthy persons was used, and as a cholesteryl ester receptor, LDL from healthy perso...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
TargetCholesteryl ester transfer protein(Homo sapiens (Human))
Chong Kun Dang Pharmaceutical
US Patent
Chong Kun Dang Pharmaceutical
US Patent
Affinity DataIC50: 3.90nMAssay Description:As a protein source for cholesteryl ester transfer, plasma from healthy persons was used, and as a cholesteryl ester receptor, LDL from healthy perso...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:The following experiment was performed to evaluate the ability of [1,2,4]triazolo[4,3-a]quinoxaline derivative of the present invention to inhibit th...More data for this Ligand-Target Pair
TargetCholesteryl ester transfer protein(Homo sapiens (Human))
Chong Kun Dang Pharmaceutical
US Patent
Chong Kun Dang Pharmaceutical
US Patent
Affinity DataIC50: 5nMAssay Description:As a protein source for cholesteryl ester transfer, plasma from healthy persons was used, and as a cholesteryl ester receptor, LDL from healthy perso...More data for this Ligand-Target Pair
TargetCholesteryl ester transfer protein(Homo sapiens (Human))
Chong Kun Dang Pharmaceutical
US Patent
Chong Kun Dang Pharmaceutical
US Patent
Affinity DataIC50: 5.30nMAssay Description:As a protein source for cholesteryl ester transfer, plasma from healthy persons was used, and as a cholesteryl ester receptor, LDL from healthy perso...More data for this Ligand-Target Pair
TargetCholesteryl ester transfer protein(Homo sapiens (Human))
Chong Kun Dang Pharmaceutical
US Patent
Chong Kun Dang Pharmaceutical
US Patent
Affinity DataIC50: 5.40nMAssay Description:As a protein source for cholesteryl ester transfer, plasma from healthy persons was used, and as a cholesteryl ester receptor, LDL from healthy perso...More data for this Ligand-Target Pair
TargetCholesteryl ester transfer protein(Homo sapiens (Human))
Chong Kun Dang Pharmaceutical
US Patent
Chong Kun Dang Pharmaceutical
US Patent
Affinity DataIC50: 5.80nMAssay Description:As a protein source for cholesteryl ester transfer, plasma from healthy persons was used, and as a cholesteryl ester receptor, LDL from healthy perso...More data for this Ligand-Target Pair
TargetCholesteryl ester transfer protein(Homo sapiens (Human))
Chong Kun Dang Pharmaceutical
US Patent
Chong Kun Dang Pharmaceutical
US Patent
Affinity DataIC50: 5.90nMAssay Description:As a protein source for cholesteryl ester transfer, plasma from healthy persons was used, and as a cholesteryl ester receptor, LDL from healthy perso...More data for this Ligand-Target Pair