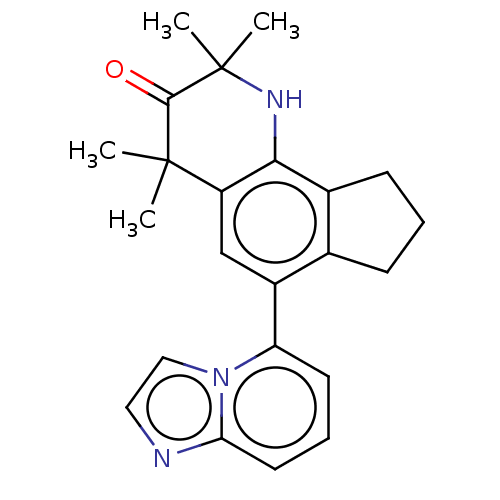
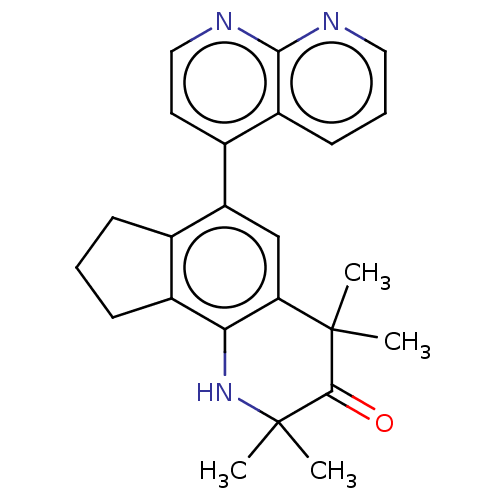
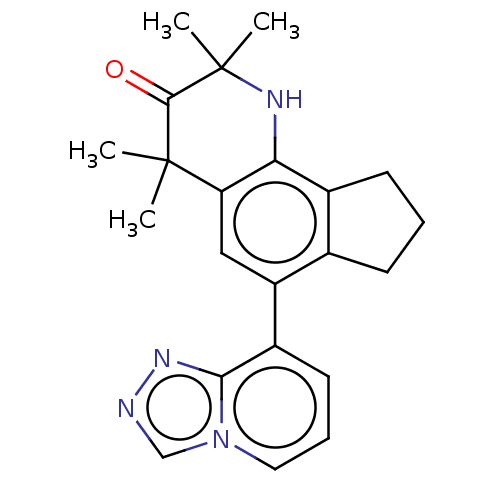
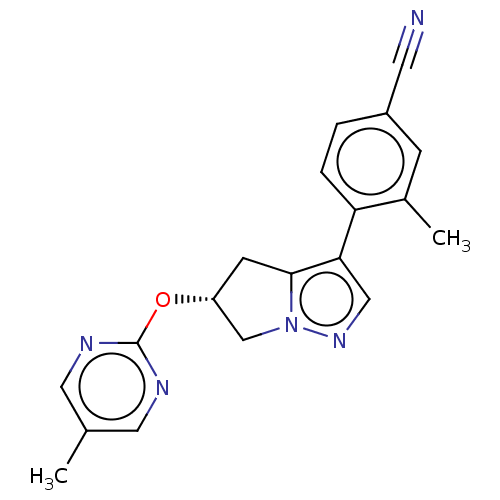
Affinity DataKi: 3.90nMAssay Description:Binding affinity to human progesterone receptor by competitive assayMore data for this Ligand-Target Pair
Affinity DataKi: 6nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 15nMAssay Description:Displacement of [3H]-progesterone from cytosolic fraction of human recombinant progesterone-B receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 15nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 16nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 22nMAssay Description:Displacement of [3H]-progesterone from cytosolic fraction of human recombinant progesterone-B receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 28nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 35nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 35nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 46nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 53nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 55nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 69nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 106nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 146nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 151nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 151nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 337nMAssay Description:Displacement of [3H]-aldosterone from cytosolic fraction of human recombinant mineralocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >384nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >384nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >384nMAssay Description:Displacement of [3H]-dexamethasone from cytosolic fraction of human recombinant glucocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 403nMAssay Description:Displacement of [3H]-progesterone from cytosolic fraction of human recombinant progesterone-B receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Displacement of [3H]-aldosterone from cytosolic fraction of human recombinant mineralocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Displacement of [3H]-aldosterone from cytosolic fraction of human recombinant mineralocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Displacement of [3H]-aldosterone from cytosolic fraction of human recombinant mineralocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 1.19E+3nMAssay Description:Displacement of [3H]-progesterone from cytosolic fraction of human recombinant progesterone-B receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >1.25E+3nMAssay Description:Displacement of [3H]-progesterone from cytosolic fraction of human recombinant progesterone-B receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >1.25E+3nMAssay Description:Displacement of [3H]-progesterone from cytosolic fraction of human recombinant progesterone-B receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >1.25E+3nMAssay Description:Displacement of [3H]-progesterone from cytosolic fraction of human recombinant progesterone-B receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >1.25E+3nMAssay Description:Displacement of [3H]-progesterone from cytosolic fraction of human recombinant progesterone-B receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >1.25E+3nMAssay Description:Displacement of [3H]-progesterone from cytosolic fraction of human recombinant progesterone-B receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 1.48E+3nMAssay Description:Displacement of [3H]-progesterone from cytosolic fraction of human recombinant progesterone-B receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 2.27E+3nMAssay Description:Displacement of [3H]-progesterone from cytosolic fraction of human recombinant progesterone-B receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >2.30E+3nMAssay Description:Displacement of fluormone ES2 from human recombinant estrogen receptor-alpha expressed in insect Sf9 cells after 120 mins by fluorescence polarizatio...More data for this Ligand-Target Pair
Affinity DataKi: >2.30E+3nMAssay Description:Displacement of fluormone ES2 from human recombinant estrogen receptor-beta expressed in insect Hi5 cells after 120 mins by fluorescence polarization...More data for this Ligand-Target Pair
Affinity DataKi: >2.30E+3nMAssay Description:Displacement of [3H]methyltrienolone from cytosolic fraction of androgen receptor in human LANCAP cells after 24 hrs by scintillation counting analys...More data for this Ligand-Target Pair
Affinity DataKi: >3.00E+3nMAssay Description:Displacement of [3H]-aldosterone from cytosolic fraction of human recombinant mineralocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >3.00E+3nMAssay Description:Displacement of [3H]-aldosterone from cytosolic fraction of human recombinant mineralocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >3.00E+3nMAssay Description:Displacement of [3H]-aldosterone from cytosolic fraction of human recombinant mineralocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: >3.33E+3nMAssay Description:Displacement of [3H]-aldosterone from cytosolic fraction of human recombinant mineralocorticoid receptor by scintillation counting analysisMore data for this Ligand-Target Pair
TargetCytochrome P450 11B2, mitochondrial(Homo sapiens (Human))
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Affinity DataIC50: 1.20nMAssay Description:Inhibition of human CYP11B2 expressed in Chinese hamster V79 cell mitochondria assessed as reduction in aldosterone production using deoxycorticoster...More data for this Ligand-Target Pair
TargetCytochrome P450 11B2, mitochondrial(Homo sapiens (Human))
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Affinity DataIC50: 2.20nMAssay Description:Inhibition of human CYP11B2 expressed in Chinese hamster V79 cell mitochondria assessed as reduction in aldosterone production using deoxycorticoster...More data for this Ligand-Target Pair
TargetCytochrome P450 11B2, mitochondrial(Homo sapiens (Human))
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Affinity DataIC50: 2.20nMAssay Description:Inhibition of human CYP11B2 expressed in Chinese hamster V79 cell mitochondria assessed as reduction in aldosterone production using deoxycorticoster...More data for this Ligand-Target Pair
TargetCytochrome P450 11B2, mitochondrial(Homo sapiens (Human))
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Affinity DataIC50: 2.30nMAssay Description:Inhibition of human CYP11B2 expressed in Chinese hamster V79 cell mitochondria assessed as reduction in aldosterone production using deoxycorticoster...More data for this Ligand-Target Pair
TargetCytochrome P450 11B2, mitochondrial(Homo sapiens (Human))
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Affinity DataIC50: 2.5nMAssay Description:Inhibition of human CYP11B2 expressed in Chinese hamster V79 cell mitochondria assessed as reduction in aldosterone production using deoxycorticoster...More data for this Ligand-Target Pair
TargetCytochrome P450 11B2, mitochondrial(Homo sapiens (Human))
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Affinity DataIC50: 3.20nMAssay Description:Inhibition of human CYP11B2 expressed in Chinese hamster V79 cell mitochondria assessed as reduction in aldosterone production using deoxycorticoster...More data for this Ligand-Target Pair
TargetCytochrome P450 11B2, mitochondrial(Homo sapiens (Human))
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Affinity DataIC50: 3.70nMAssay Description:Inhibition of human CYP11B2 expressed in Chinese hamster V79 cell mitochondria assessed as reduction in aldosterone production using deoxycorticoster...More data for this Ligand-Target Pair
TargetCytochrome P450 11B2, mitochondrial(Homo sapiens (Human))
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:Inhibition of human CYP11B2 expressed in Chinese hamster V79 cell mitochondria assessed as reduction in aldosterone production using deoxycorticoster...More data for this Ligand-Target Pair
TargetCytochrome P450 11B2, mitochondrial(Homo sapiens (Human))
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Mitsubishi Tanabe Pharma
Curated by ChEMBL
Affinity DataIC50: 4.20nMAssay Description:Inhibition of human CYP11B2 expressed in Chinese hamster V79 cell mitochondria assessed as reduction in aldosterone production using deoxycorticoster...More data for this Ligand-Target Pair


 3D Structure (crystal)
3D Structure (crystal)