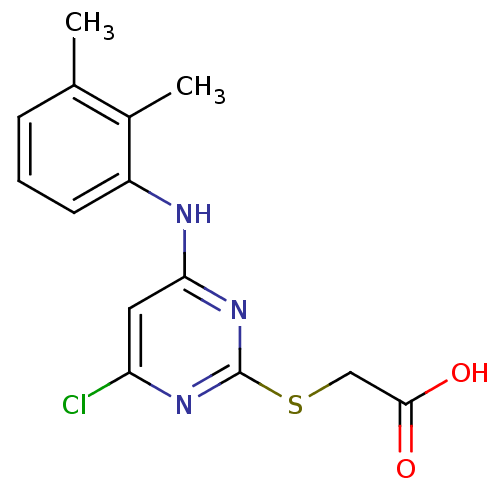
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Eberhard Karls University Tuebingen
Eberhard Karls University Tuebingen
Affinity DatapH: 7.4 T: 2°CAssay Description:For assays of intact cells, 5000,000 freshly isolated PMNL cells were resuspended in PGC buffer. After preincubation with the test compounds for 15 m...More data for this Ligand-Target Pair
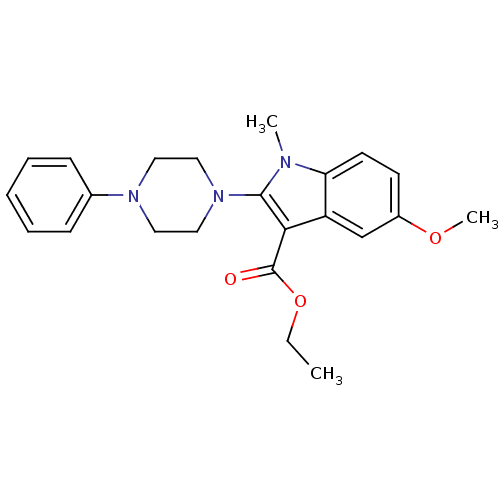
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Eberhard Karls University Tuebingen
Eberhard Karls University Tuebingen
Affinity DatapH: 7.4 T: 2°CAssay Description:Purified 5-LO was added to reaction mix containing the test compounds. For efficient inhibition of 5-LO, low hydroperoxide levels are important. Glut...More data for this Ligand-Target Pair
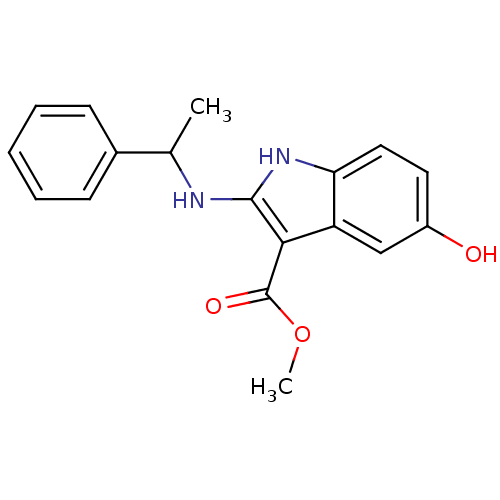
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Eberhard Karls University Tuebingen
Eberhard Karls University Tuebingen
Affinity DatapH: 7.4 T: 2°CAssay Description:Purified 5-LO was added to reaction mix containing the test compounds. For efficient inhibition of 5-LO, low hydroperoxide levels are important. Glut...More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Eberhard Karls University Tuebingen
Eberhard Karls University Tuebingen
Affinity DatapH: 7.4 T: 2°CAssay Description:Purified 5-LO was added to reaction mix containing the test compounds. For efficient inhibition of 5-LO, low hydroperoxide levels are important. Glut...More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Eberhard Karls University Tuebingen
Eberhard Karls University Tuebingen
Affinity DatapH: 7.4 T: 2°CAssay Description:Purified 5-LO was added to reaction mix containing the test compounds. For efficient inhibition of 5-LO, low hydroperoxide levels are important. Glut...More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Eberhard Karls University Tuebingen
Eberhard Karls University Tuebingen
Affinity DatapH: 7.4 T: 2°CAssay Description:Purified 5-LO was added to reaction mix containing the test compounds. For efficient inhibition of 5-LO, low hydroperoxide levels are important. Glut...More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
Eberhard Karls University Tuebingen
Eberhard Karls University Tuebingen
Affinity DatapH: 7.4 T: 2°CAssay Description:Purified 5-LO was added to reaction mix containing the test compounds. For efficient inhibition of 5-LO, low hydroperoxide levels are important. Glut...More data for this Ligand-Target Pair