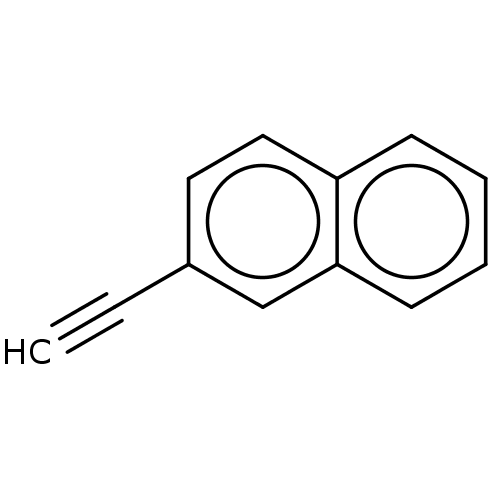
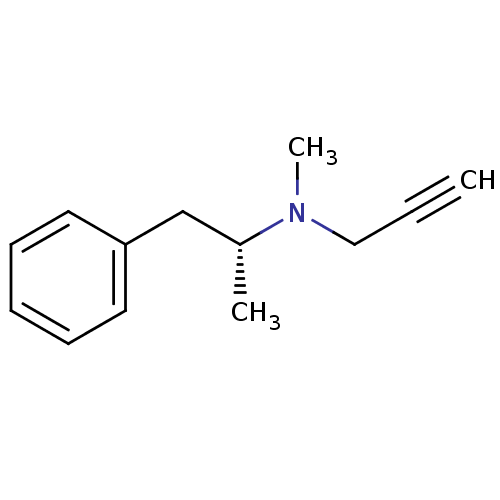
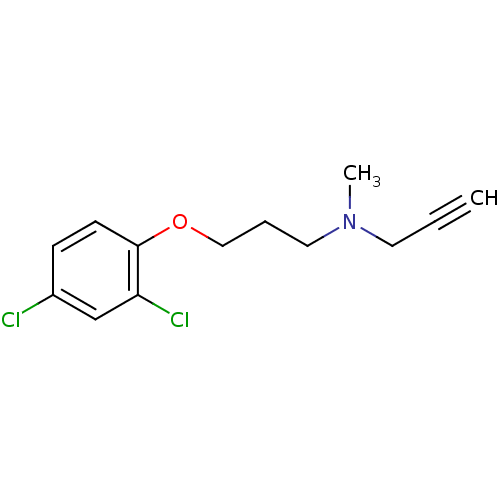
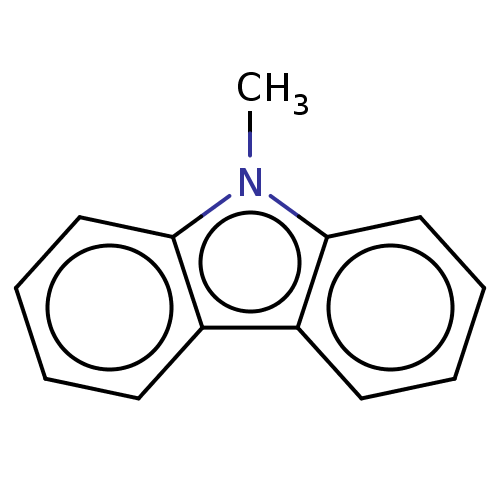
Affinity DataKi: 80nMAssay Description:Mechanism based inhibition of rat cytochrome P450 CYP2B1 measured by 7-ethoxycoumarin O-deethylase activityMore data for this Ligand-Target Pair
Affinity DataKi: 140nMAssay Description:Mechanism based inhibition of rat cytochrome P450 CYP2B1 measured by 7-pentoxyresorufin O-deethylation activity (PROD)More data for this Ligand-Target Pair
Affinity DataKi: 800nMAssay Description:Mechanism based inhibition of rat cytochrome P450 CYP2B1 measured by 7-pentoxyresorufin O-deethylation activity (PROD)More data for this Ligand-Target Pair
Affinity DataKi: 1.05E+3nMAssay Description:Mechanism based inhibition of rat cytochrome P450 CYP2B1 measured by 7-EFC O-deethylation activityMore data for this Ligand-Target Pair
Affinity DataKi: 1.50E+3nMAssay Description:Mechanism based inhibition of rat cytochrome P450 CYP2B1 measured by 7-EFC O-deethylation activityMore data for this Ligand-Target Pair
Affinity DataKi: 2.40E+3nMAssay Description:Mechanism based inhibition of rat cytochrome P450 2B1More data for this Ligand-Target Pair
Affinity DataKi: 5.20E+3nMAssay Description:Mechanism based inhibition of rat cytochrome P450 CYP2B1More data for this Ligand-Target Pair
Affinity DataKi: 5.80E+3nMAssay Description:Mechanism based inhibition of rat cytochrome P450 CYP2B1 measured by 7-EFC O-deethylation activityMore data for this Ligand-Target Pair
Affinity DataKi: 1.10E+4nMAssay Description:Mechanism based inhibition of rat cytochrome P450 2B1More data for this Ligand-Target Pair
Affinity DataKi: 1.20E+4nMAssay Description:Mechanism based inhibition of rat cytochrome P450 2B1More data for this Ligand-Target Pair
Affinity DataKi: 3.10E+4nMAssay Description:Mechanism based inhibition of rat cytochrome P450 2B1More data for this Ligand-Target Pair