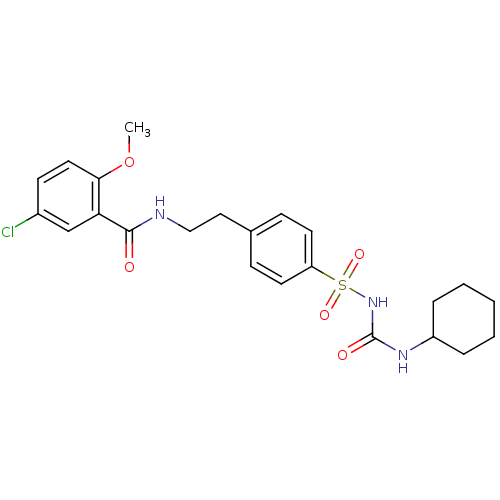
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 240nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 480nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 480nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.40E+3nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 2.00E+3nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 4.20E+3nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >3.00E+4nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 4.50E+4nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair
TargetATP synthase subunit gamma, mitochondrial(Bos taurus)
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 4.70E+5nMAssay Description:Inhibitory concentration towards rat mitochondrial F1F0 ATP hydrolase using a pyruvate kinase / lactate dehydrogenase systemMore data for this Ligand-Target Pair