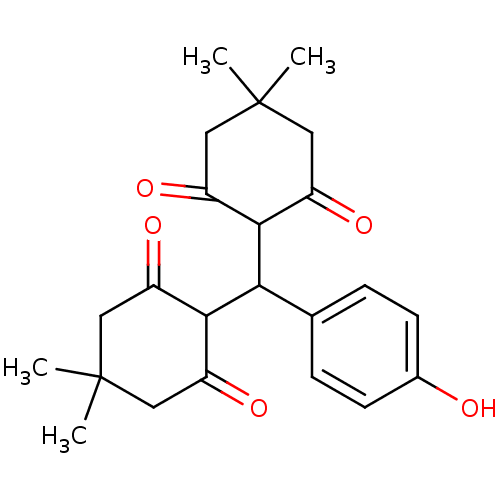
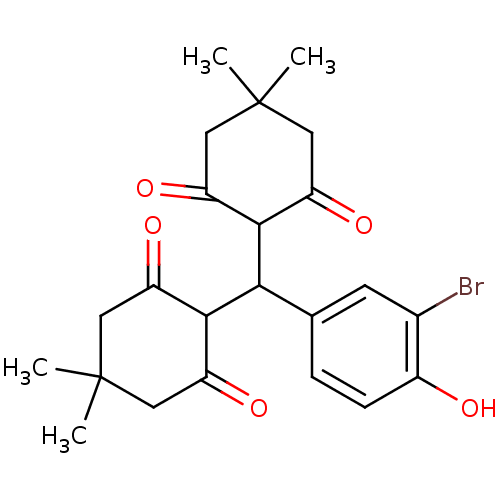
Affinity DataIC50: 7.80E+3nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 1.25E+4nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 1.63E+4nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 1.75E+4nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 2.13E+4nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 2.64E+4nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 2.71E+4nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 2.84E+4nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 3.08E+4nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 3.30E+4nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 3.31E+4nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 3.54E+4nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 3.97E+4nMpH: 8.0 T: 2°CAssay Description:Lipoxygenase inhibiting activity was measured by slightly modifying the spectrometric method. Lipoxygenase type I-B and linoleic acid were purchased...More data for this Ligand-Target Pair
Affinity DataIC50: 4.04E+4nMpH: 8.0 T: 2°CAssay Description:Lipoxygenase inhibiting activity was measured by slightly modifying the spectrometric method. Lipoxygenase type I-B and linoleic acid were purchased...More data for this Ligand-Target Pair
Affinity DataIC50: 4.19E+4nMpH: 8.0 T: 2°CAssay Description:Lipoxygenase inhibiting activity was measured by slightly modifying the spectrometric method. Lipoxygenase type I-B and linoleic acid were purchased...More data for this Ligand-Target Pair
Affinity DataIC50: 4.43E+4nMpH: 8.0 T: 2°CAssay Description:Lipoxygenase inhibiting activity was measured by slightly modifying the spectrometric method. Lipoxygenase type I-B and linoleic acid were purchased...More data for this Ligand-Target Pair
Affinity DataIC50: 4.73E+4nMpH: 8.0 T: 2°CAssay Description:Lipoxygenase inhibiting activity was measured by slightly modifying the spectrometric method. Lipoxygenase type I-B and linoleic acid were purchased...More data for this Ligand-Target Pair
Affinity DataIC50: 4.96E+4nMpH: 8.0 T: 2°CAssay Description:Lipoxygenase inhibiting activity was measured by slightly modifying the spectrometric method. Lipoxygenase type I-B and linoleic acid were purchased...More data for this Ligand-Target Pair
Affinity DataIC50: 5.15E+4nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 5.25E+4nMpH: 8.0 T: 2°CAssay Description:Lipoxygenase inhibiting activity was measured by slightly modifying the spectrometric method. Lipoxygenase type I-B and linoleic acid were purchased...More data for this Ligand-Target Pair
Affinity DataIC50: 5.29E+4nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 1.91E+5nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 1.96E+5nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataIC50: 1.98E+5nMpH: 8.0 T: 2°CAssay Description:In vitro liposygenase inhibition assay activity was measured by modifying the spectrophotometric method developed by Tappel. The compound was prepar...More data for this Ligand-Target Pair
Affinity DataKi: 5.76E+4nM ΔG°: -24.2kJ/molepH: 8.0 T: 2°CAssay Description:In vitro lipoxygenase inhibition assay, lipoxygenase inhibiting activity was convenintly measured by slightly modifying the spectrometic method devel...More data for this Ligand-Target Pair
Affinity DataKi: 7.17E+4nM ΔG°: -23.7kJ/molepH: 8.0 T: 2°CAssay Description:In vitro lipoxygenase inhibition assay, lipoxygenase inhibiting activity was convenintly measured by slightly modifying the spectrometic method devel...More data for this Ligand-Target Pair