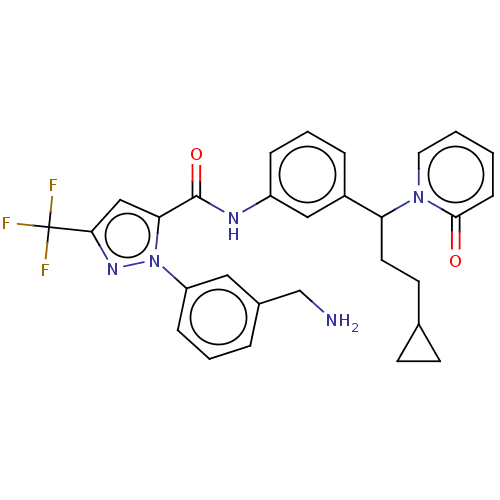
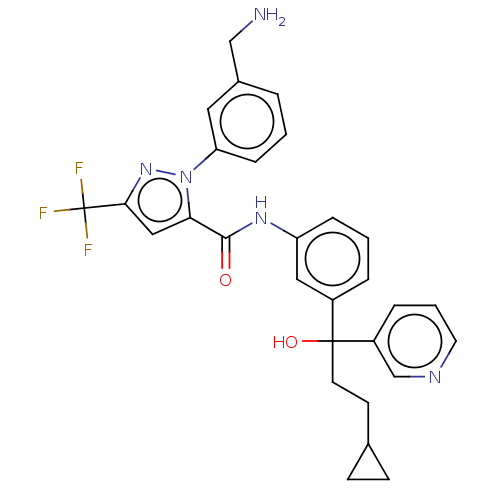
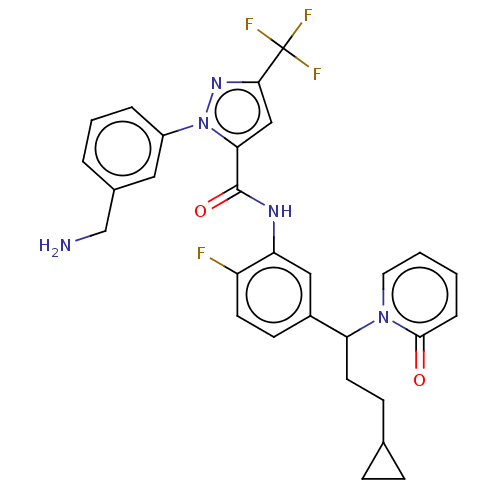
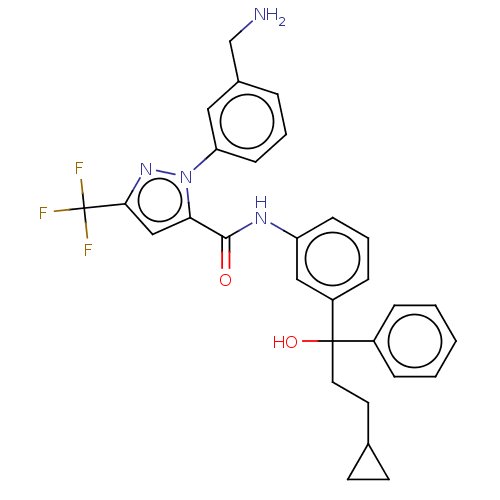
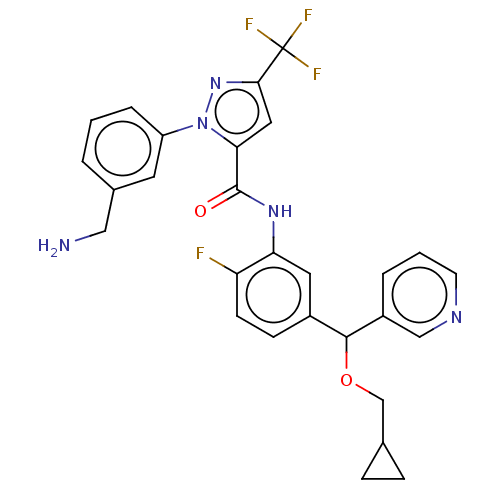
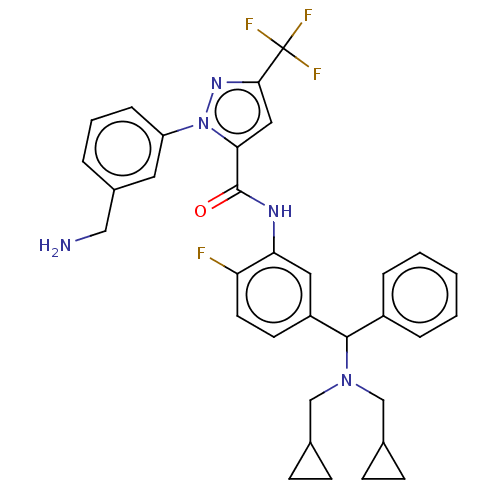
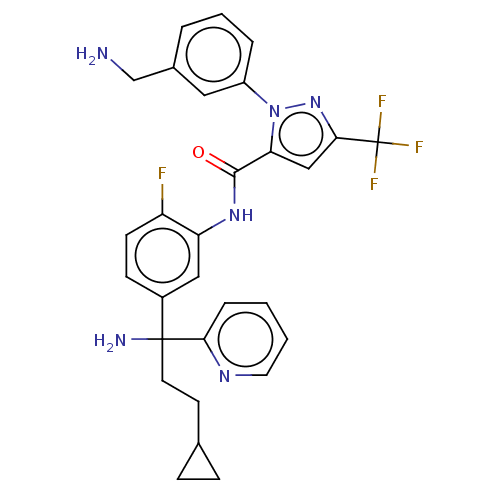
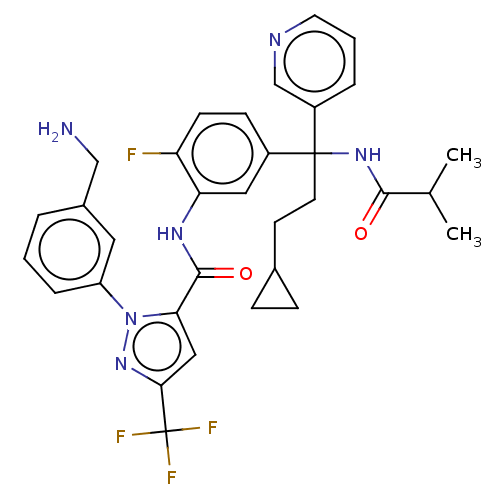
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair
Affinity DataKi: 25.1nMAssay Description:The effect of compounds of the invention on human plasma kallikrein activity was determined using the chromogenic substrates. In these experiments, 2...More data for this Ligand-Target Pair