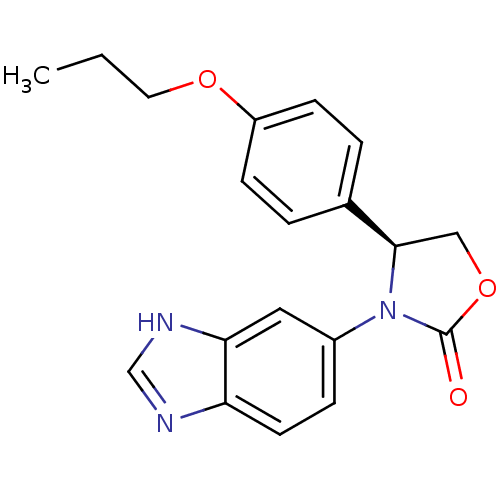
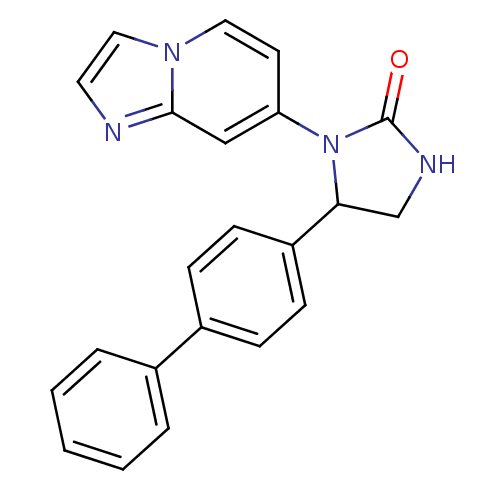
Affinity DataKi: 12.5nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataKi: 33.6nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataKi: 37.4nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataKi: 40.8nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataKi: 53.5nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataKi: 58.8nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataKi: 62.5nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataKi: 75nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataKi: 149nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataKi: 7.25E+3nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataIC50: 160nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataIC50: 170nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataIC50: 238nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataIC50: 240nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataIC50: 254nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataIC50: 297nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataIC50: 397nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataIC50: 482nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataIC50: 882nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair
Affinity DataIC50: 3.02E+4nMAssay Description:All measurements were performed with a BioAssay Reader HTS-7000Plus for microplates (Perkin Elmer) at 30� C. QC activity was evaluated fluorometrical...More data for this Ligand-Target Pair