Affinity DataIC50: 316nMAssay Description:Inhibition of human sPLA2X using 1,2-bis(heptanoylthio) glycerophosphocholine substrate incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+4nMAssay Description:Binding affinity to sPLA2X (unknown origin) by NMR spectroscopy based displacement assayMore data for this Ligand-Target Pair

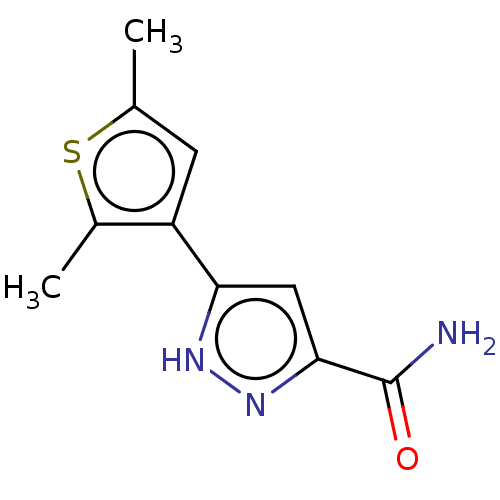
 3D Structure (crystal)
3D Structure (crystal)