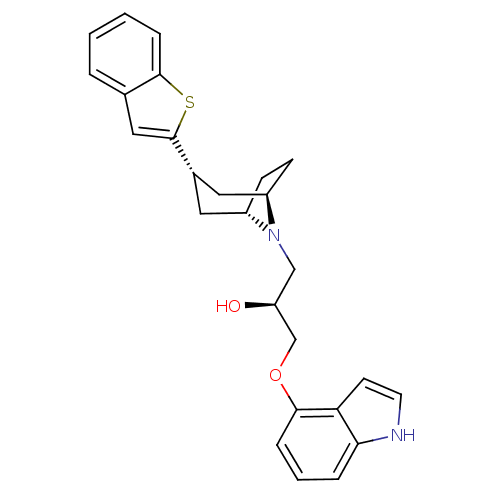
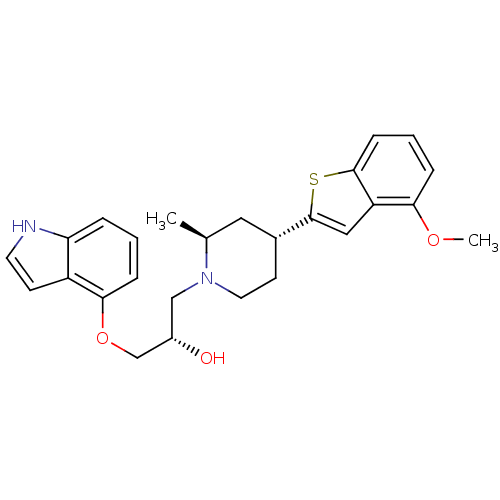
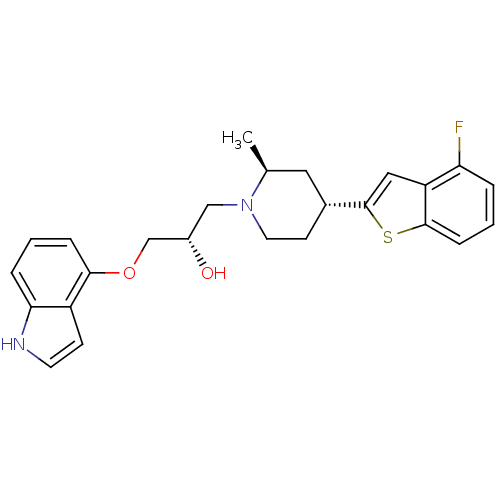
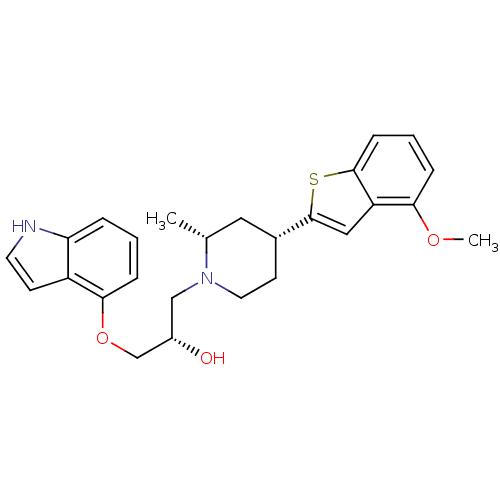
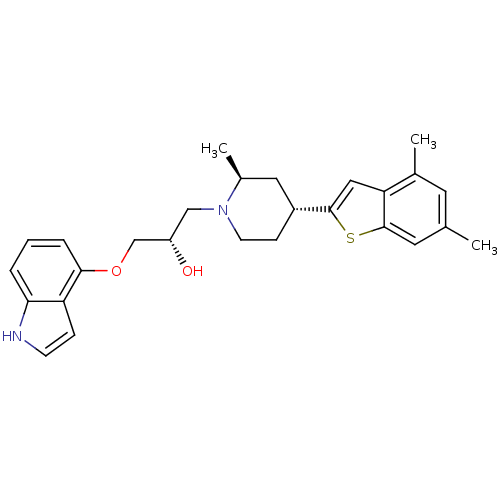
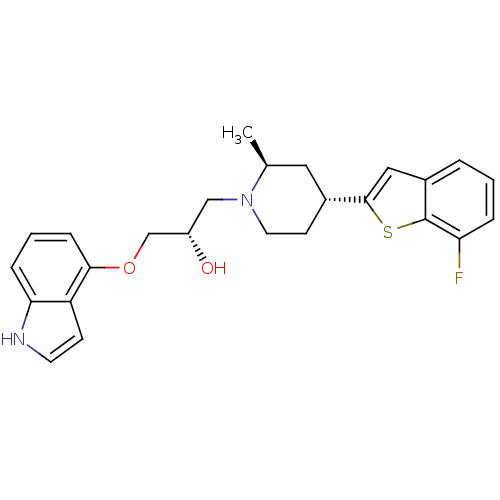
Affinity DataKi: 0.0700nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 0.170nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 0.170nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 0.270nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 0.310nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 0.390nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 0.440nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 0.510nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 0.580nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 0.690nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 0.980nMAssay Description:Inhibition of 5-HT induced [35S]-GTP-gammaS, binding at human cloned 5-HT 1A receptors.More data for this Ligand-Target Pair
Affinity DataKi: 0.990nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 1.80nMAssay Description:Inhibition of 5-HT induced [35S]-GTP-gammaS, binding at human cloned 5-HT 1A receptors.More data for this Ligand-Target Pair
Affinity DataKi: 1.80nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 1.90nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 2.20nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 2.30nMAssay Description:Inhibition of 5-HT induced [35S]-GTP-gammaS, binding at human cloned 5-HT 1A receptors.More data for this Ligand-Target Pair
Affinity DataKi: 2.80nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 3nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 3.10nMAssay Description:Inhibition of 5-HT induced [35S]-GTP-gammaS, binding at human cloned 5-HT 1A receptors.More data for this Ligand-Target Pair
Affinity DataKi: 3.30nMAssay Description:Inhibition of 5-HT induced [35S]-GTP-gammaS, binding at human cloned 5-HT 1A receptors.More data for this Ligand-Target Pair
Affinity DataKi: 3.5nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 3.5nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 3.5nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 3.60nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 4.20nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 4.20nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 4.90nMAssay Description:Inhibition of 5-HT induced [35S]-GTP-gammaS, binding at human cloned 5-HT 1A receptors.More data for this Ligand-Target Pair
Affinity DataKi: 5.30nMAssay Description:Inhibition of 5-HT induced [35S]-GTP-gammaS, binding at human cloned 5-HT 1A receptors.More data for this Ligand-Target Pair
Affinity DataKi: 5.5nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 5.70nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 6.40nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 6.90nMAssay Description:Inhibition of 5-HT induced [35S]-GTP-gammaS, binding at human cloned 5-HT 1A receptors.More data for this Ligand-Target Pair
Affinity DataKi: 7.5nMAssay Description:Inhibition of 5-HT induced [35S]-GTP-gammaS, binding at human cloned 5-HT 1A receptors.More data for this Ligand-Target Pair
Affinity DataKi: 8.30nMAssay Description:Inhibition of 5-HT induced [35S]-GTP-gammaS, binding at human cloned 5-HT 1A receptors.More data for this Ligand-Target Pair
Affinity DataKi: 8.40nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 8.5nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 8.80nMAssay Description:Inhibition of 5-HT induced [35S]-GTP-gammaS, binding at human cloned 5-HT 1A receptors.More data for this Ligand-Target Pair
Affinity DataKi: 8.80nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:In vitro affinity of the compound at the 5-HT reuptake site using [3H]-paroxetine as radioligand in rat frontal cortex membranesMore data for this Ligand-Target Pair
Affinity DataKi: 12nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 12nMAssay Description:Inhibition of 5-HT induced [35S]-GTP-gammaS, binding at human cloned 5-HT 1A receptors.More data for this Ligand-Target Pair
Affinity DataKi: 14nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 14nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair
Affinity DataKi: 14nMAssay Description:In vitro binding affinity at 5-hydroxytryptamine 1A receptor by [3H]-8-OH-DPAT displacement.More data for this Ligand-Target Pair