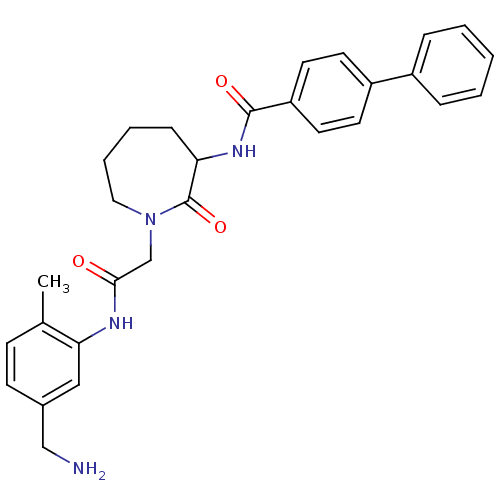
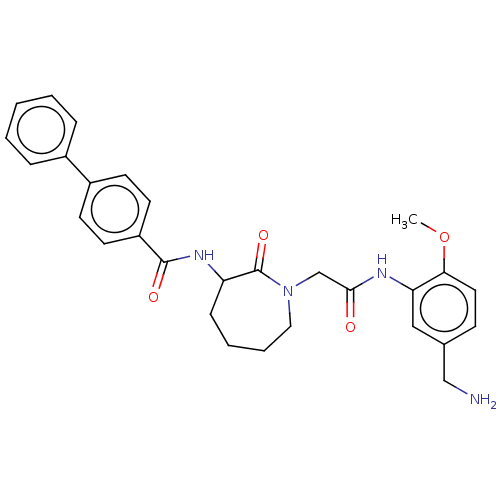
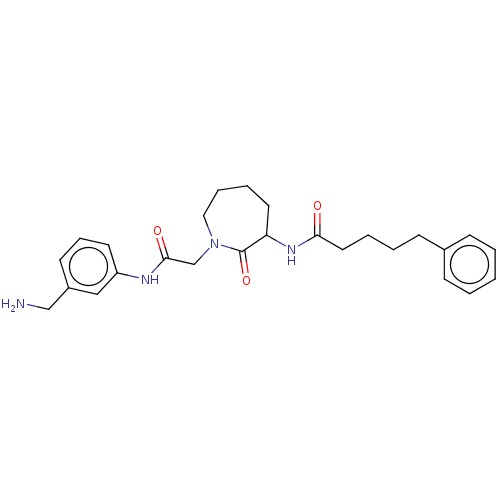
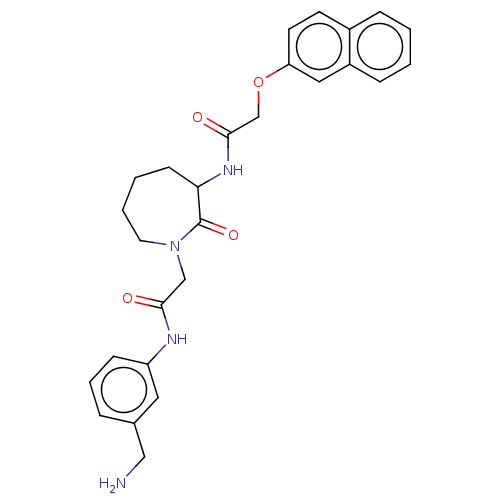
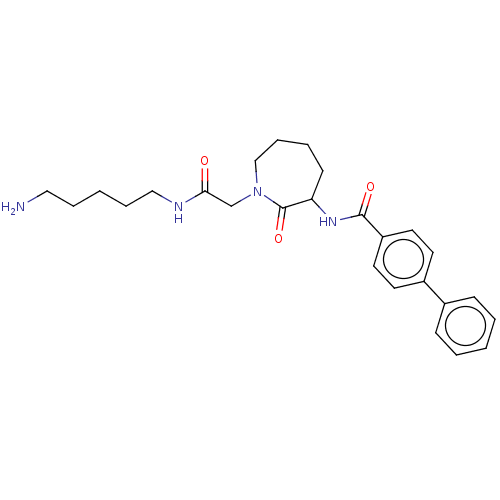
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 38nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 74nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetVitamin K-dependent protein C(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >130nMAssay Description:Compound was tested for inhibition of human activated protein C (APC)More data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >130nMAssay Description:Compound was tested for inhibition of human alpha-thrombin (FIIa)More data for this Ligand-Target Pair
TargetTissue-type plasminogen activator(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >130nMAssay Description:Compound was tested for inhibition of human tissue type plasminogen activatorMore data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >130nMAssay Description:Compound was tested for inhibition of human urokinase-type plasminogen activatorMore data for this Ligand-Target Pair
TargetSerine protease 1(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >130nMAssay Description:Compound was tested for inhibition of human trypsinMore data for this Ligand-Target Pair
TargetPlasminogen(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >130nMAssay Description:Compound was tested for inhibition of human plasminMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >130nMAssay Description:Compound was tested for inhibition of human Coagulation factor XMore data for this Ligand-Target Pair
TargetSerine protease 1(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >170nMAssay Description:Compound was tested for inhibition of human trypsinMore data for this Ligand-Target Pair
TargetTissue-type plasminogen activator(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >170nMAssay Description:Compound was tested for inhibition of human tissue type plasminogen activatorMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >170nMAssay Description:Compound was tested for inhibition of human Coagulation factor XMore data for this Ligand-Target Pair
TargetPlasminogen(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >170nMAssay Description:Compound was tested for inhibition of human plasminMore data for this Ligand-Target Pair
TargetVitamin K-dependent protein C(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >170nMAssay Description:Compound was tested for inhibition of human activated protein C (APC)More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >170nMAssay Description:Compound was tested for inhibition of human urokinase-type plasminogen activatorMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >170nMAssay Description:Compound was tested for inhibition of human alpha-thrombin (FIIa)More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 200nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 250nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >250nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetPlasminogen(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 330nMAssay Description:Compound was tested for inhibition of human plasminMore data for this Ligand-Target Pair
TargetSerine protease 1(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 360nMAssay Description:Compound was tested for inhibition of human trypsinMore data for this Ligand-Target Pair
TargetSerine protease 1(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >450nMAssay Description:Compound was tested for inhibition of human trypsinMore data for this Ligand-Target Pair
TargetPlasminogen(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >450nMAssay Description:Compound was tested for inhibition of human plasminMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >450nMAssay Description:Compound was tested for inhibition of human Coagulation factor XMore data for this Ligand-Target Pair
TargetTissue-type plasminogen activator(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >450nMAssay Description:Compound was tested for inhibition of human tissue type plasminogen activatorMore data for this Ligand-Target Pair
TargetVitamin K-dependent protein C(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >450nMAssay Description:Compound was tested for inhibition of human activated protein C (APC)More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >450nMAssay Description:Compound was tested for inhibition of human urokinase-type plasminogen activatorMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >450nMAssay Description:Compound was tested for inhibition of human alpha-thrombin (FIIa)More data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >870nMAssay Description:Compound was tested for inhibition of human Coagulation factor XMore data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >870nMAssay Description:Compound was tested for inhibition of human urokinase-type plasminogen activatorMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >870nMAssay Description:Compound was tested for inhibition of human alpha-thrombin (FIIa)More data for this Ligand-Target Pair
TargetVitamin K-dependent protein C(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >870nMAssay Description:Compound was tested for inhibition of human activated protein C (APC)More data for this Ligand-Target Pair
TargetTissue-type plasminogen activator(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >870nMAssay Description:Compound was tested for inhibition of human tissue type plasminogen activatorMore data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 890nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 980nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 980nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.30E+3nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 2.10E+3nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 2.90E+3nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 2.90E+3nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 3.60E+3nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 4.60E+3nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 4.60E+3nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 5.20E+3nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.10E+4nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.20E+4nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.20E+4nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.30E+4nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.50E+4nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair
TargetTryptase beta-2/delta/gamma(Homo sapiens (Human))
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: >3.30E+4nMAssay Description:Inhibition of human tryptase.More data for this Ligand-Target Pair