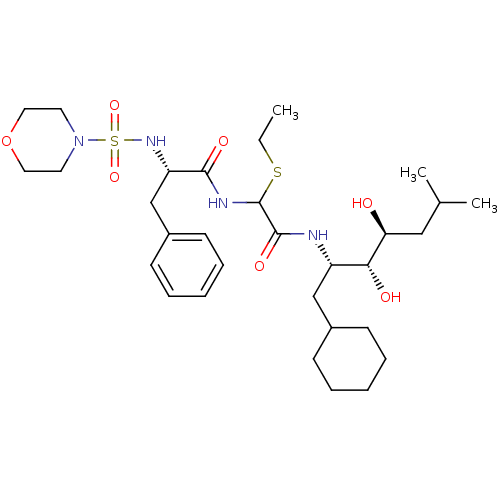
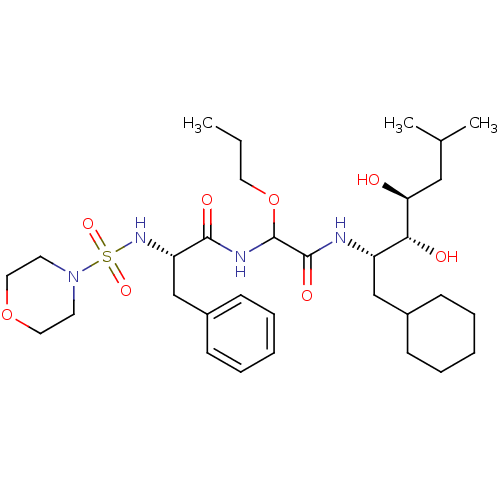
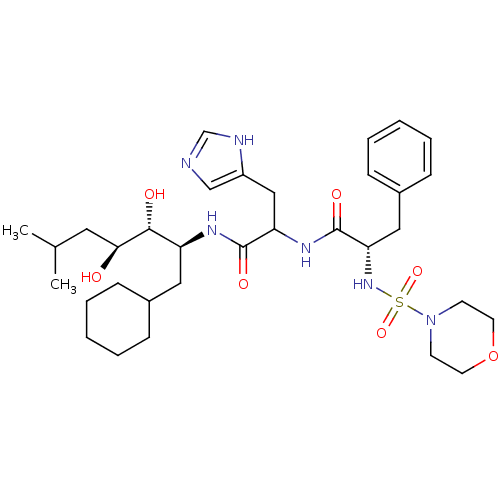
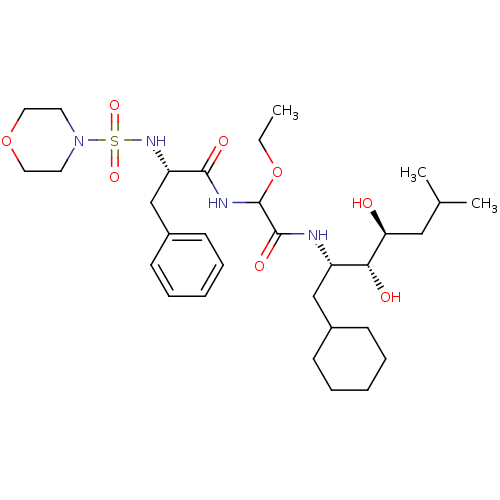
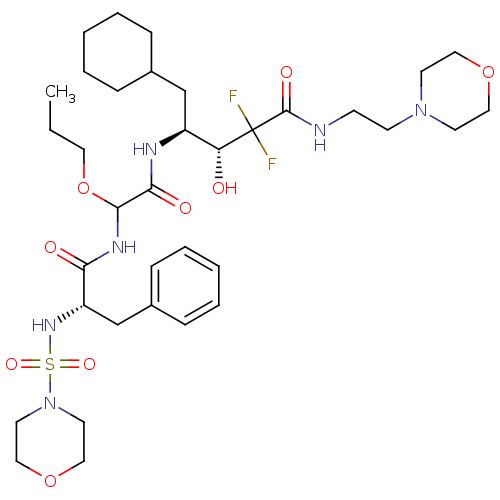
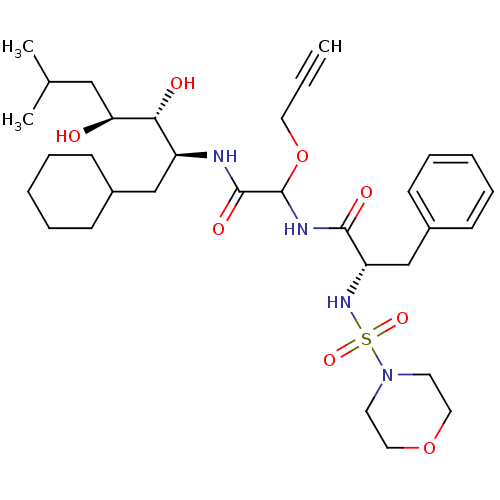
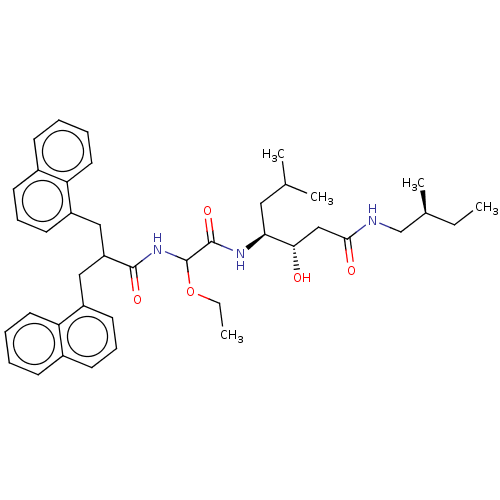
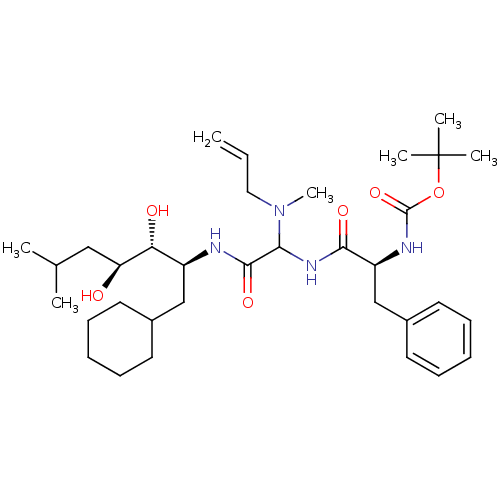
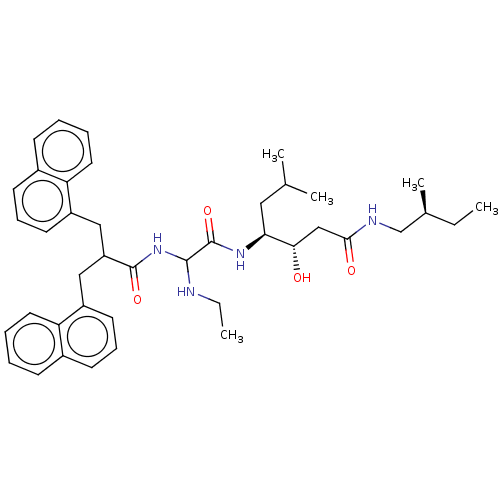
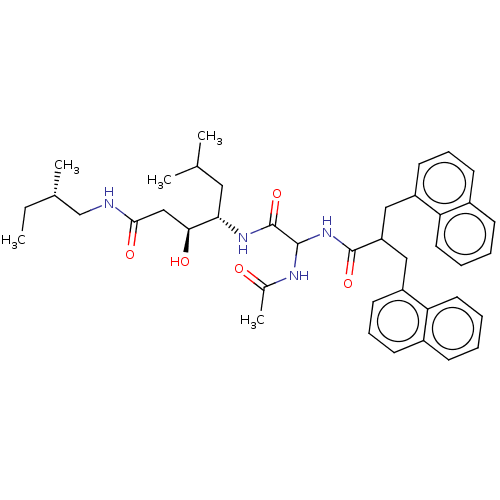
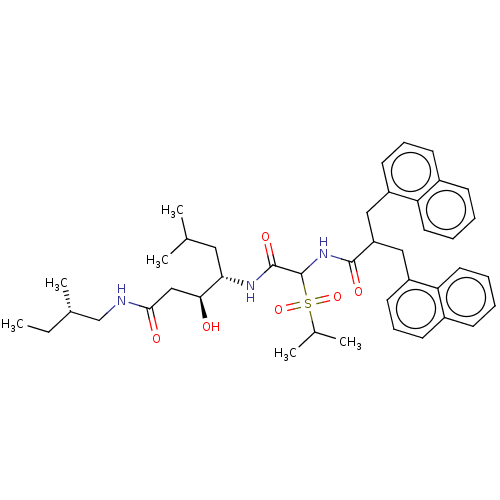
Affinity DataIC50: 0.130nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 0.170nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 0.180nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 0.210nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 0.220nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 0.230nMAssay Description:Inhibitory activity against monkey reninMore data for this Ligand-Target Pair
Affinity DataIC50: 0.25nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 0.440nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 0.520nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 1.40nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 2.10nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibitory activity against monkey reninMore data for this Ligand-Target Pair
Affinity DataIC50: 5.30nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Inhibitory activity against bovine cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Inhibitory activity against bovine cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:Inhibitory activity against bovine cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:Inhibitory activity against bovine cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: 19nMAssay Description:Inhibitory activity against bovine cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: 21nMAssay Description:Inhibitory activity against bovine cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: 22nMAssay Description:Inhibitory activity against bovine cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: 26nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 28nMAssay Description:Inhibitory activity against bovine cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: 35nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 60nMAssay Description:Inhibitory activity against bovine cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: 74nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 80nMAssay Description:Inhibitory activity against bovine cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: 85nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 95nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 95nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 131nMAssay Description:Inhibitory activity against bovine cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: 180nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 210nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 310nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 470nMAssay Description:Inhibitory activity against bovine cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: 500nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 516nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 615nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 633nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 656nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 699nMAssay Description:Inhibitory activity against bovine cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: 936nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+3nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+3nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+3nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+3nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+3nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+3nMAssay Description:Inhibitory activity against monkey renin in vitro.More data for this Ligand-Target Pair