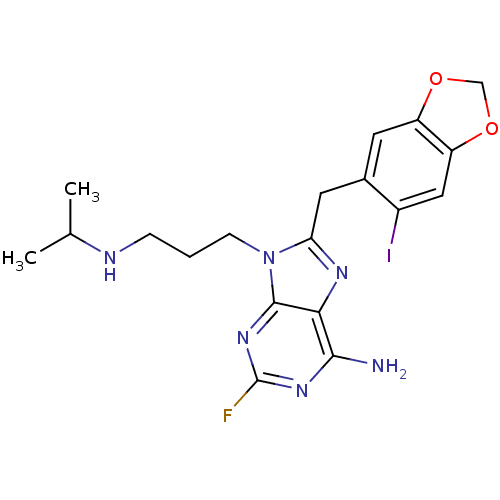
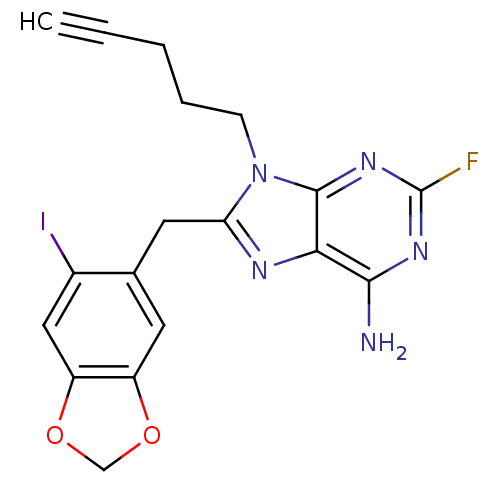
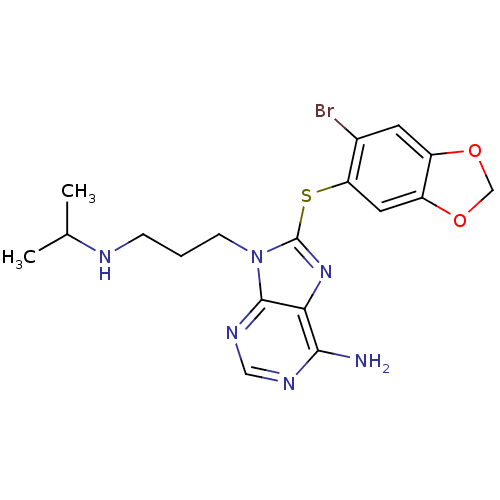
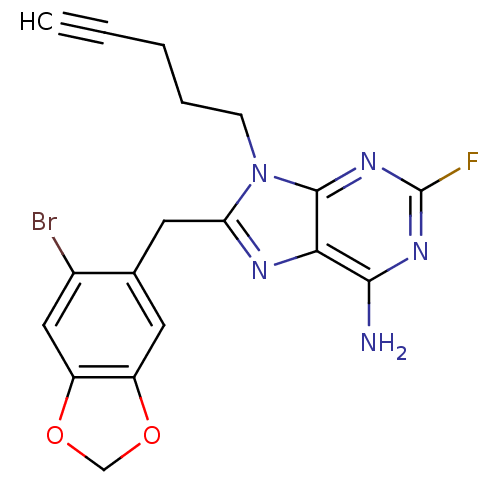
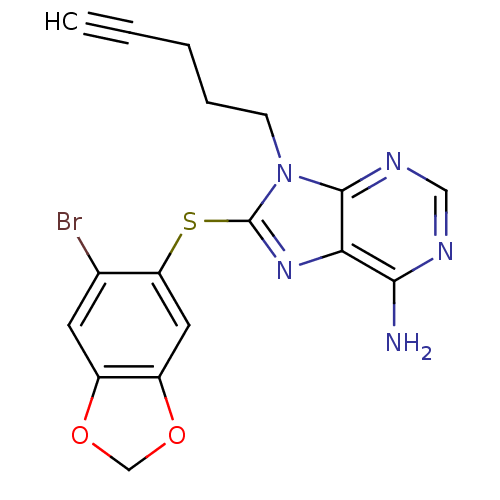
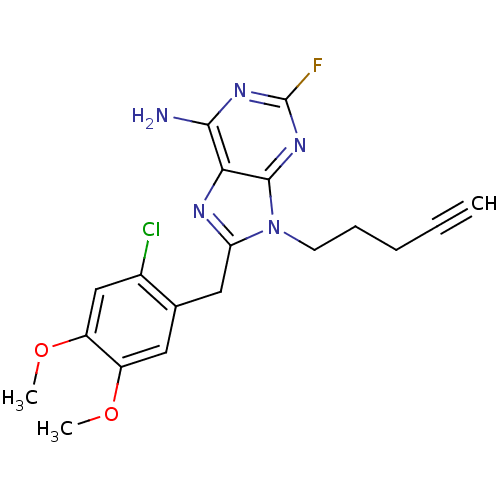
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataIC50: 50nMAssay Description:Inhibition of Her2 degradation in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataIC50: 80nMAssay Description:Inhibition of Her2 degradation in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataIC50: 90nMAssay Description:Inhibition of Her2 degradation in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:Inhibition of Her2 degradation in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataIC50: 205nMAssay Description:Inhibition of Her2 degradation in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataIC50: 210nMAssay Description:Inhibition of Her2 degradation in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataIC50: 300nMAssay Description:Inhibition of Her2 degradation in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataIC50: 365nMAssay Description:Inhibition of Her2 degradation in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataIC50: 3.00E+4nMAssay Description:Inhibition of Her2 degradation in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataIC50: >5.00E+4nMAssay Description:Inhibition of Her2 degradation in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataIC50: >5.00E+4nMAssay Description:Inhibition of Her2 degradation in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 30.5nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer MDA-MB-468 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 77.1nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer MDA-MB-468 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 39.1nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer MDA-MB-468 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 38.8nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 56.5nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 2.40E+3nMAssay Description:Binding affinity to Hsp90 in lung tissueMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: >1.50E+4nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 6.00E+3nMAssay Description:Binding affinity to Hsp90 in heart tissueMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 4.60E+3nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 77.2nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 55.4nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer MDA-MB-468 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 50.4nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 5.20E+3nMAssay Description:Binding affinity to Hsp90 in lung tissueMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 4.50E+3nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer MDA-MB-468 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 16.1nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 22.3nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: >1.50E+4nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 10.8nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 23.7nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer MDA-MB-468 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 2.20E+3nMAssay Description:Binding affinity to Hsp90 in lung tissueMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 10.9nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer MDA-MB-468 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 1.33E+4nMAssay Description:Binding affinity to Hsp90 in heart tissueMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 50.4nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer SKBr3 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 10.2nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer MDA-MB-468 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 51.2nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer MDA-MB-468 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: >1.50E+4nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer MDA-MB-468 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: >1.50E+4nMAssay Description:Inhibitory activity against Hsp90 in human breast cancer MDA-MB-468 cell lineMore data for this Ligand-Target Pair
TargetHeat shock protein HSP 90-beta(Homo sapiens (Human))
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Memorial Sloan-Kettering Cancer Center
Curated by ChEMBL
Affinity DataEC50: 6.90E+3nMAssay Description:Binding affinity to Hsp90 in heart tissueMore data for this Ligand-Target Pair

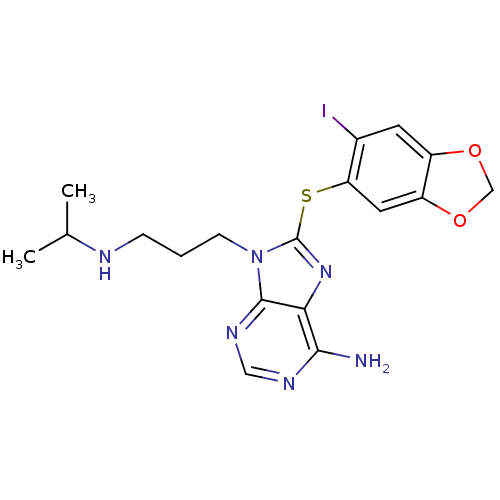
 3D Structure (crystal)
3D Structure (crystal)