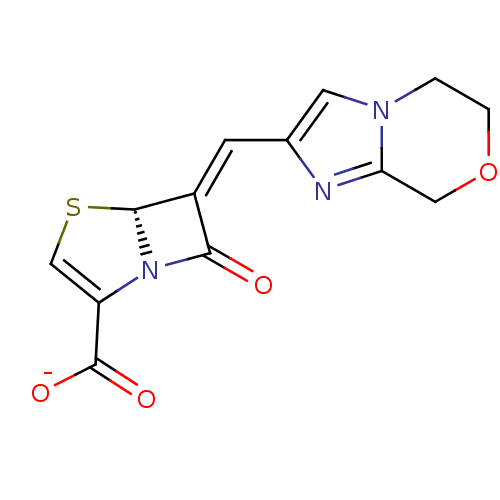
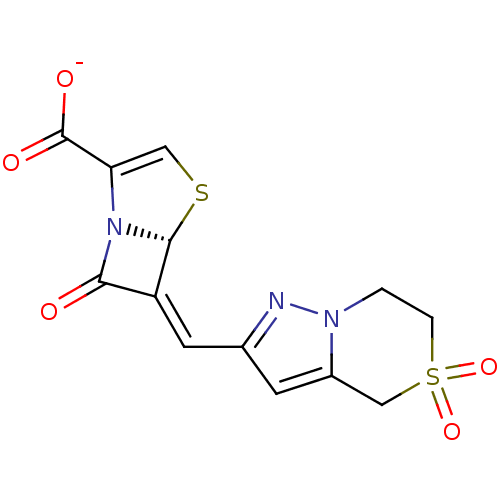
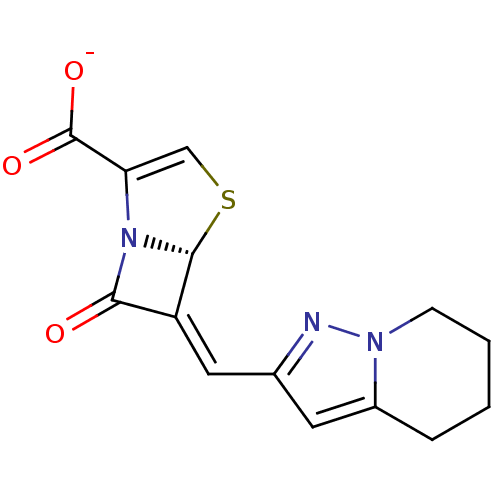
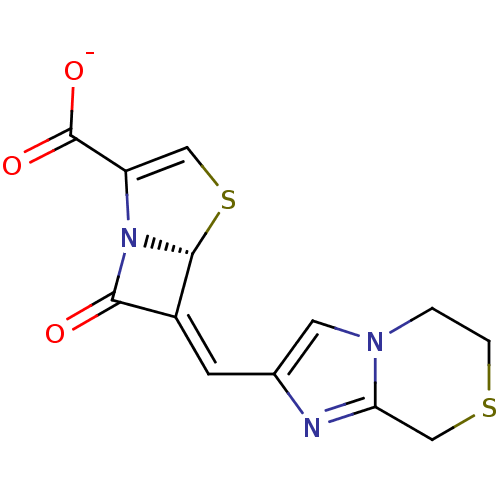
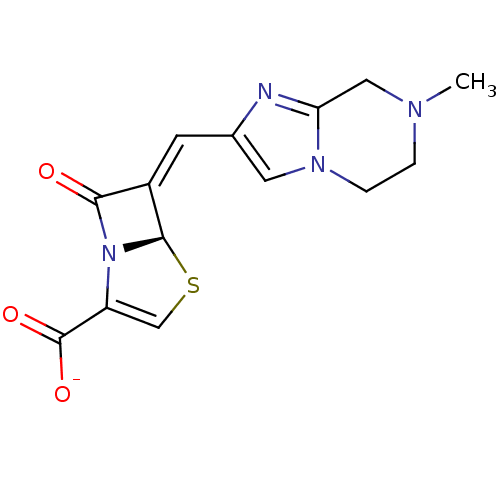
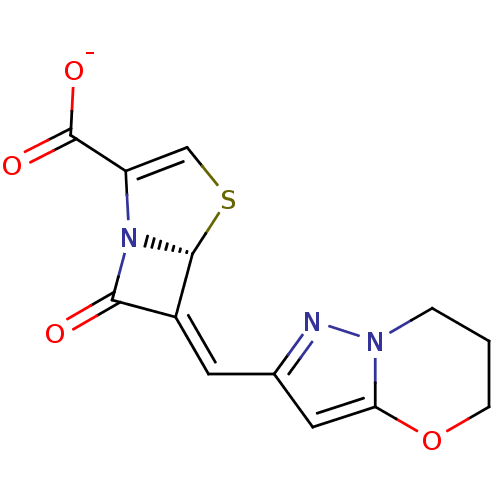
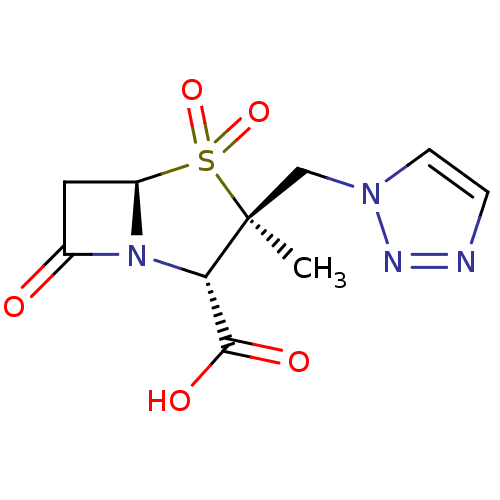
Affinity DataIC50: 0.400nMAssay Description:Inhibition of Escherichia coli TEM1More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of Escherichia coli TEM1More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of Escherichia coli TEM1More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of Enterobacter cloacae AmpCMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of Escherichia coli TEM1More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of Escherichia coli TEM1More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of Escherichia coli TEM1More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of Enterobacter cloacae AmpCMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of Escherichia coli TEM1More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of Enterobacter cloacae AmpCMore data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Inhibition of Enterobacter cloacae AmpCMore data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Inhibition of Enterobacter cloacae AmpCMore data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Inhibition of Escherichia coli TEM1More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:Inhibition of Escherichia coli TEM1More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:Inhibition of Enterobacter cloacae AmpCMore data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:Inhibition of Escherichia coli TEM1More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of Enterobacter cloacae AmpCMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of Enterobacter cloacae AmpCMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of Escherichia coli TEM1More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Inhibition of Enterobacter cloacae AmpCMore data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Inhibition of Escherichia coli TEM1More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Inhibition of Enterobacter cloacae AmpCMore data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Inhibition of Enterobacter cloacae AmpCMore data for this Ligand-Target Pair
Affinity DataIC50: 8nMAssay Description:Inhibition of Enterobacter cloacae Imi1More data for this Ligand-Target Pair
Affinity DataIC50: 8nMAssay Description:Inhibition of Bacteroides fragilis CcrAMore data for this Ligand-Target Pair
Affinity DataIC50: 9nMAssay Description:Inhibition of Enterobacter cloacae Imi1More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Inhibition of Escherichia coli TEM1More data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:Inhibition of Bacteroides fragilis CcrAMore data for this Ligand-Target Pair
Affinity DataIC50: 15nMAssay Description:Inhibition of Enterobacter cloacae AmpCMore data for this Ligand-Target Pair
Affinity DataIC50: 18nMAssay Description:Inhibition of Enterobacter cloacae Imi1More data for this Ligand-Target Pair
Affinity DataIC50: 18nMAssay Description:Inhibition of Enterobacter cloacae AmpCMore data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:Inhibition of Enterobacter cloacae Imi1More data for this Ligand-Target Pair
Affinity DataIC50: 22nMAssay Description:Inhibition of Enterobacter cloacae Imi1More data for this Ligand-Target Pair
Affinity DataIC50: 27nMAssay Description:Inhibition of Enterobacter cloacae Imi1More data for this Ligand-Target Pair
Affinity DataIC50: 28nMAssay Description:Inhibition of Enterobacter cloacae Imi1More data for this Ligand-Target Pair
Affinity DataIC50: 30nMAssay Description:Inhibition of Bacteroides fragilis CcrAMore data for this Ligand-Target Pair
Affinity DataIC50: 40nMAssay Description:Inhibition of Enterobacter cloacae Imi1More data for this Ligand-Target Pair
Affinity DataIC50: 43nMAssay Description:Inhibition of Bacteroides fragilis CcrAMore data for this Ligand-Target Pair
Affinity DataIC50: 56nMAssay Description:Inhibition of Enterobacter cloacae Imi1More data for this Ligand-Target Pair
Affinity DataIC50: 61nMAssay Description:Inhibition of Bacteroides fragilis CcrAMore data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:Inhibition of Bacteroides fragilis CcrAMore data for this Ligand-Target Pair
Affinity DataIC50: 66nMAssay Description:Inhibition of Bacteroides fragilis CcrAMore data for this Ligand-Target Pair
Affinity DataIC50: 78nMAssay Description:Inhibition of Bacteroides fragilis CcrAMore data for this Ligand-Target Pair
Affinity DataIC50: 90nMAssay Description:Inhibition of Enterobacter cloacae Imi1More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:Inhibition of Escherichia coli TEM1More data for this Ligand-Target Pair
Affinity DataIC50: 110nMAssay Description:Inhibition of Bacteroides fragilis CcrAMore data for this Ligand-Target Pair
Affinity DataIC50: 120nMAssay Description:Inhibition of Bacteroides fragilis CcrAMore data for this Ligand-Target Pair
Affinity DataIC50: 123nMAssay Description:Inhibition of Enterobacter cloacae Imi1More data for this Ligand-Target Pair
Affinity DataIC50: 140nMAssay Description:Inhibition of Bacteroides fragilis CcrAMore data for this Ligand-Target Pair
Affinity DataIC50: 162nMAssay Description:Inhibition of Bacteroides fragilis CcrAMore data for this Ligand-Target Pair