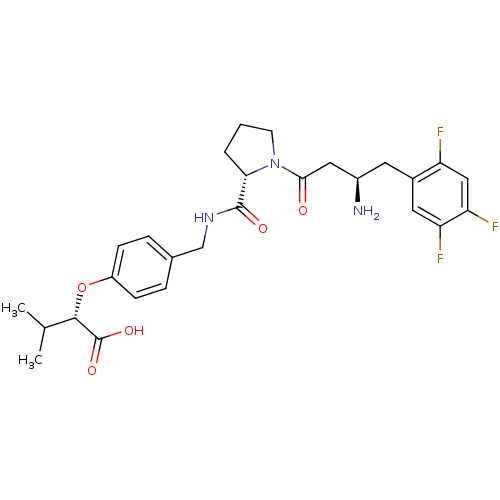
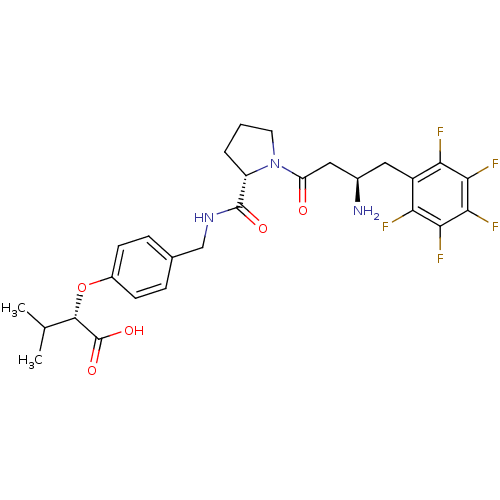
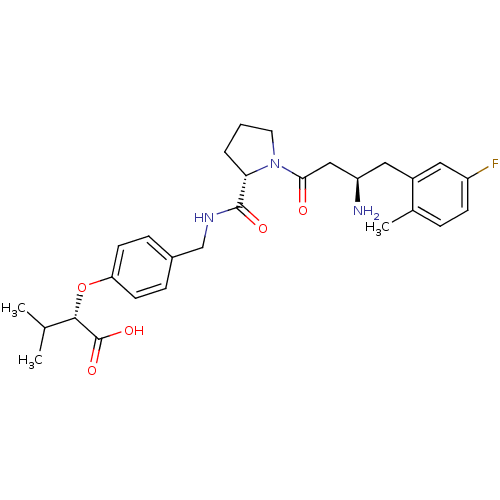
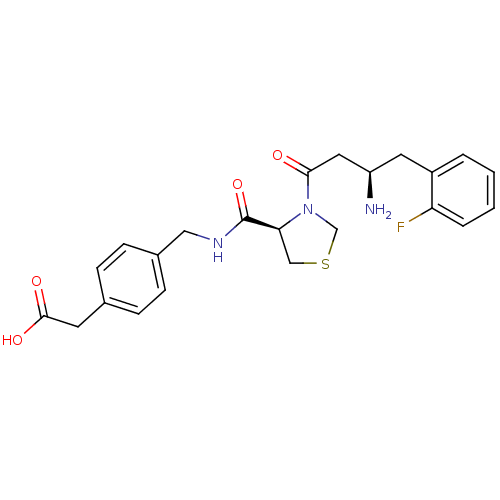
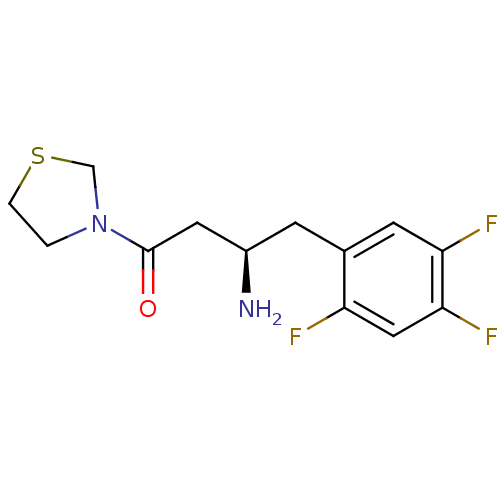
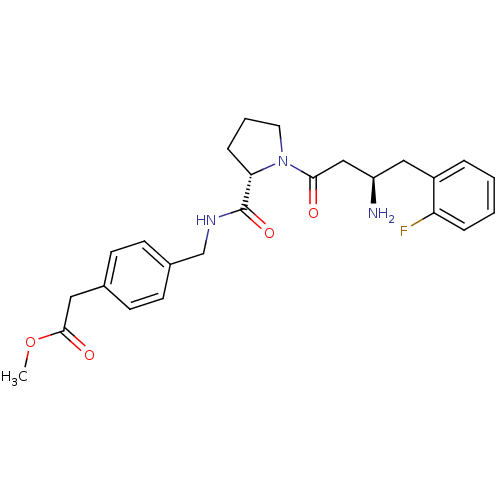
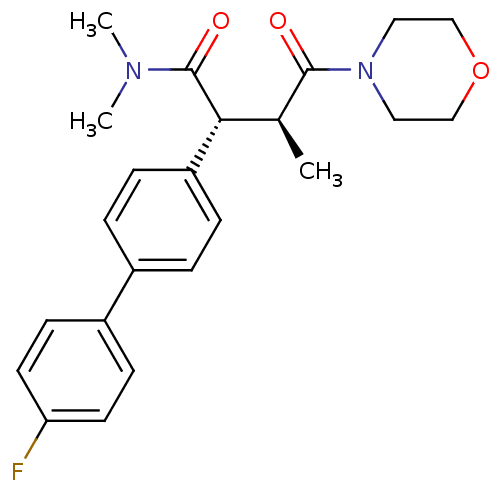
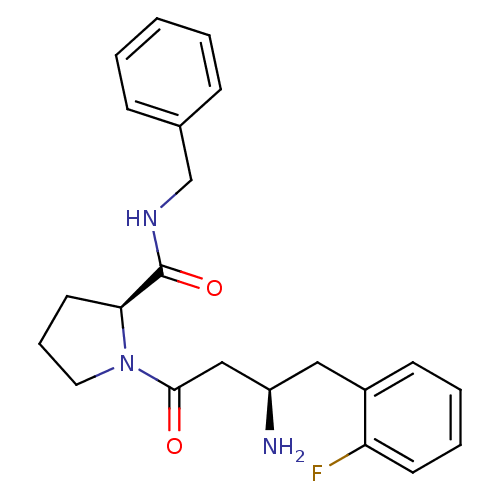
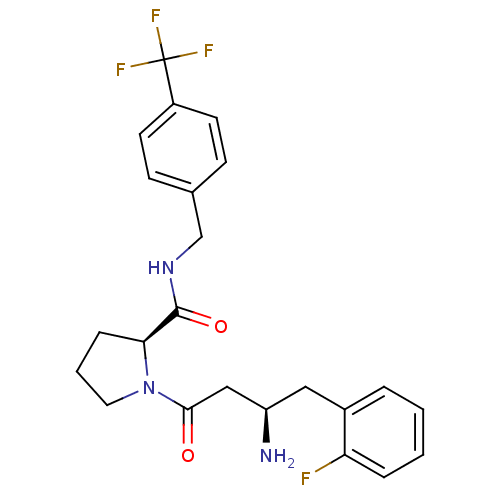
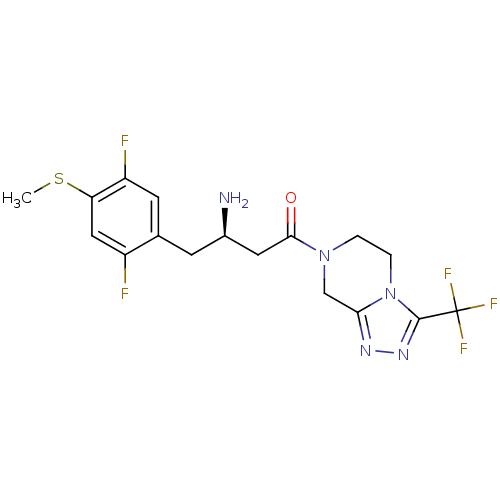
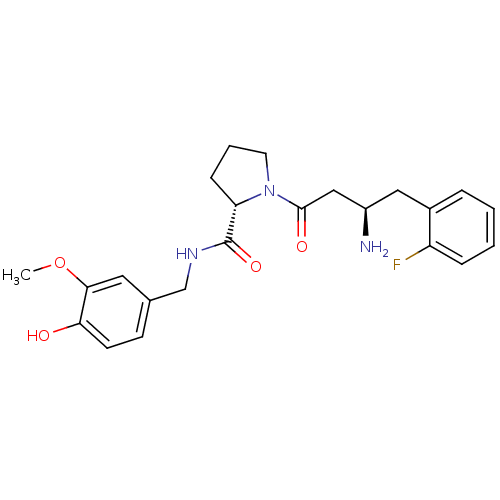
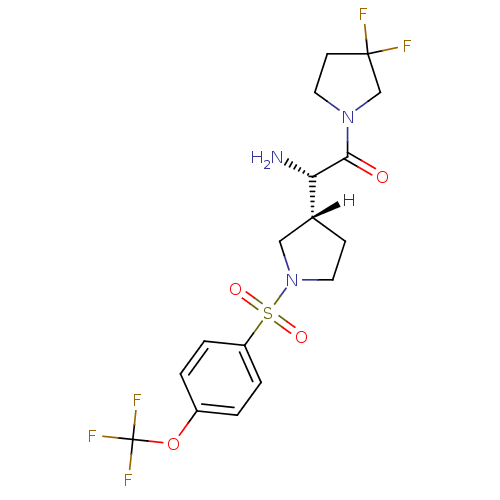
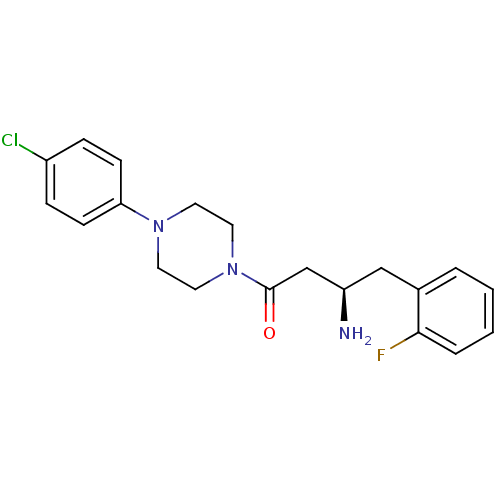
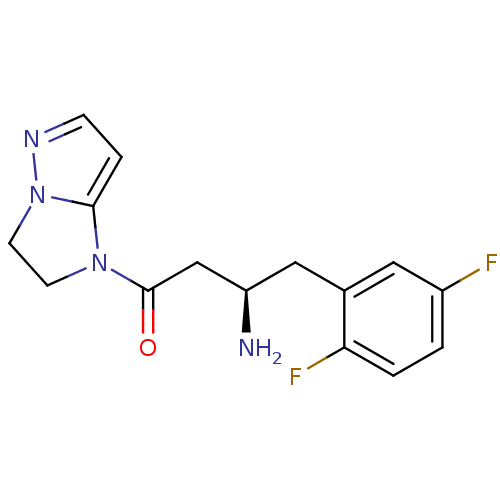
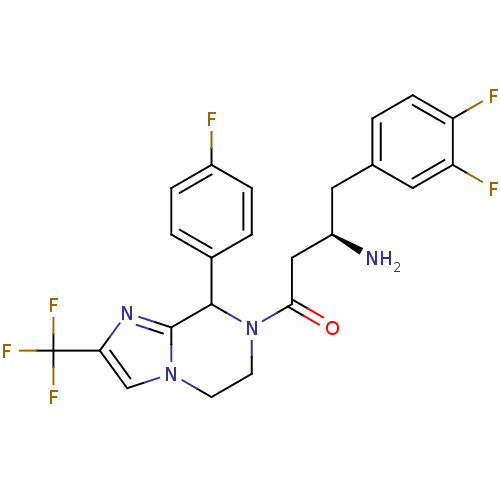
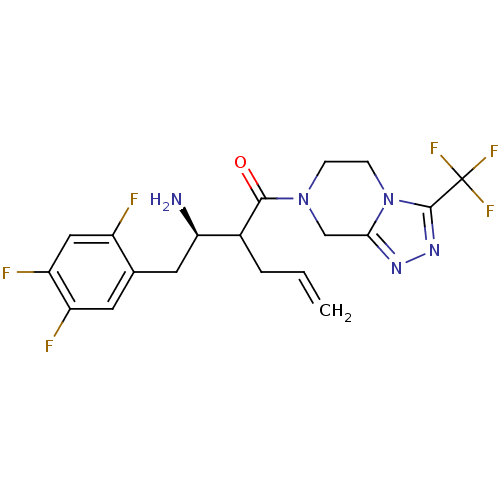
Affinity DataIC50: 2nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 8nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 21nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 33nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 37nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 119nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 170nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 310nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 520nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 539nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 731nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 780nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 864nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 995nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 1.25E+3nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 1.33E+3nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 1.40E+3nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 1.46E+3nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair
Affinity DataIC50: 2.21E+3nMpH: 7.5 T: 2°CAssay Description:The enzyme activity resulted in the formation of the fluorescent product amidomethylcoumarin (AMC), which was monitored by excitation at 360 nm and m...More data for this Ligand-Target Pair