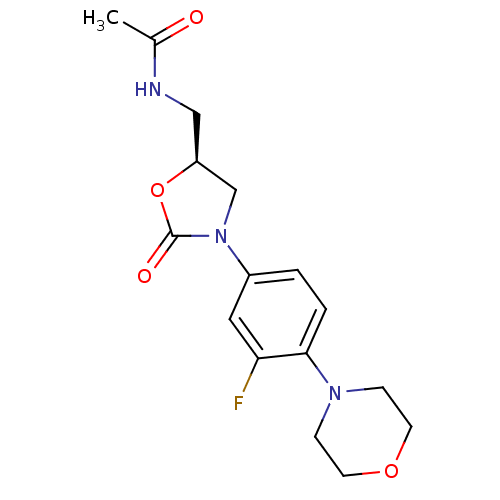
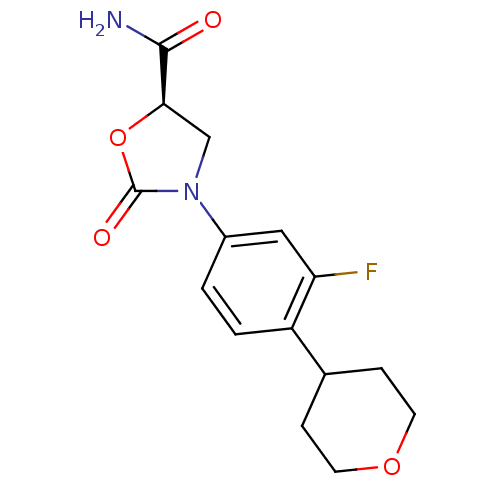
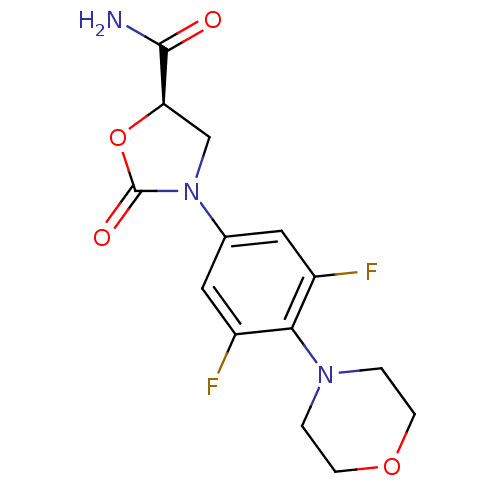
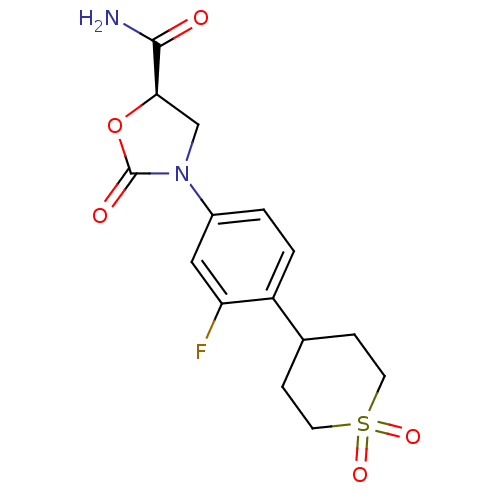
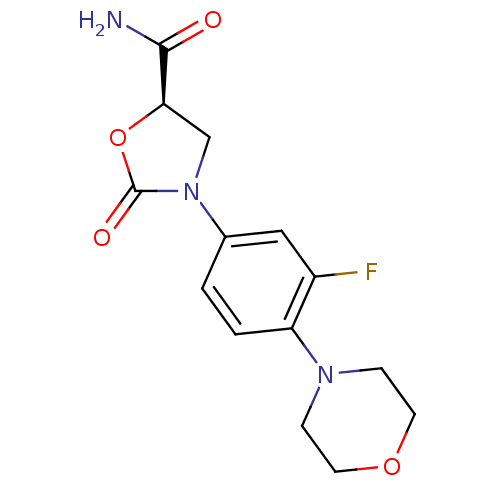
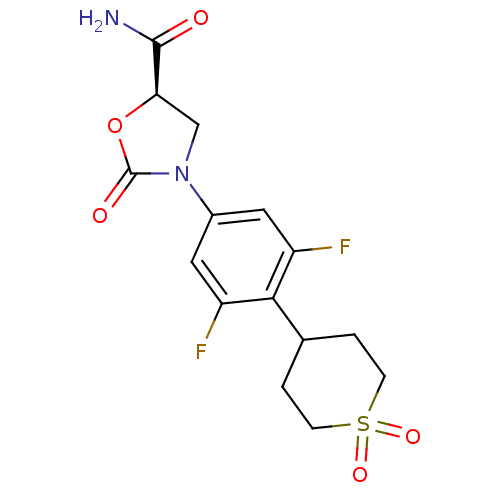
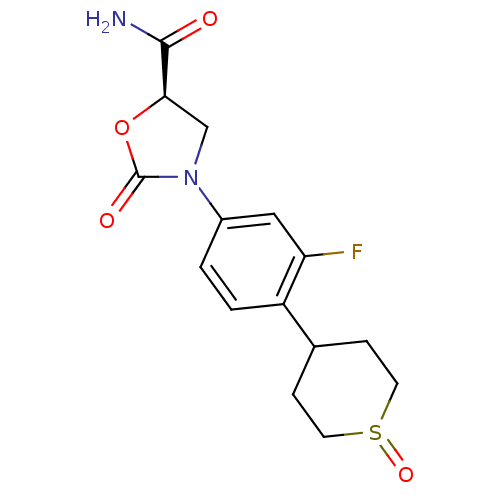
Affinity DataKi: 5.30E+4nMAssay Description:Inhibition of human recombinant MAOAMore data for this Ligand-Target Pair
Affinity DataKi: 5.80E+4nMAssay Description:Inhibition of human recombinant MAOAMore data for this Ligand-Target Pair
Affinity DataKi: 6.30E+4nMAssay Description:Inhibition of human recombinant MAOAMore data for this Ligand-Target Pair
Affinity DataKi: 1.05E+5nMAssay Description:Inhibition of human recombinant MAOAMore data for this Ligand-Target Pair
Affinity DataKi: 1.46E+5nMAssay Description:Inhibition of human recombinant MAOAMore data for this Ligand-Target Pair
Affinity DataKi: 2.93E+5nMAssay Description:Inhibition of human recombinant MAOAMore data for this Ligand-Target Pair
Affinity DataKi: 3.55E+5nMAssay Description:Inhibition of human recombinant MAOAMore data for this Ligand-Target Pair
Affinity DataIC50: >9.00E+5nMAssay Description:Inhibition of human liver microsome CYP2D6More data for this Ligand-Target Pair
Affinity DataIC50: >9.00E+5nMAssay Description:Inhibition of human liver microsome CYP1A2More data for this Ligand-Target Pair
Affinity DataIC50: >9.00E+5nMAssay Description:Inhibition of human liver microsome CYP2C19More data for this Ligand-Target Pair
Affinity DataIC50: >9.00E+5nMAssay Description:Inhibition of human liver microsome CYP2C9More data for this Ligand-Target Pair