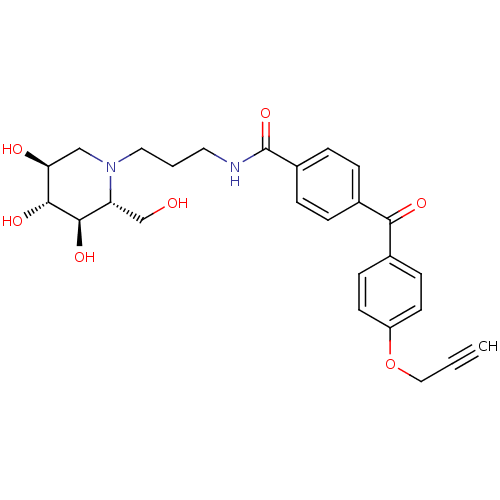
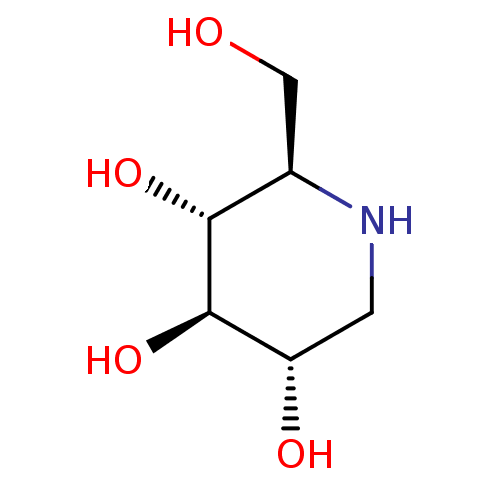
Affinity DataIC50: 20nMAssay Description:Displacement of 4-methylumbelliferyl from non-lysosomal GBA2 by fluorometryMore data for this Ligand-Target Pair
Affinity DataIC50: 75nMAssay Description:Displacement of 4-methylumbelliferyl from non-lysosomal GBA2 by fluorometryMore data for this Ligand-Target Pair
Affinity DataIC50: 350nMAssay Description:Displacement of 4-methylumbelliferyl from human GBA by fluorometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.65E+3nMAssay Description:Displacement of 4-methylumbelliferyl from human GBA by fluorometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.88E+4nMAssay Description:Displacement of 4-methylumbelliferyl from non-lysosomal GBA2 by fluorometryMore data for this Ligand-Target Pair
Affinity DataIC50: 5.06E+5nMAssay Description:Displacement of 4-methylumbelliferyl from human GBA by fluorometryMore data for this Ligand-Target Pair