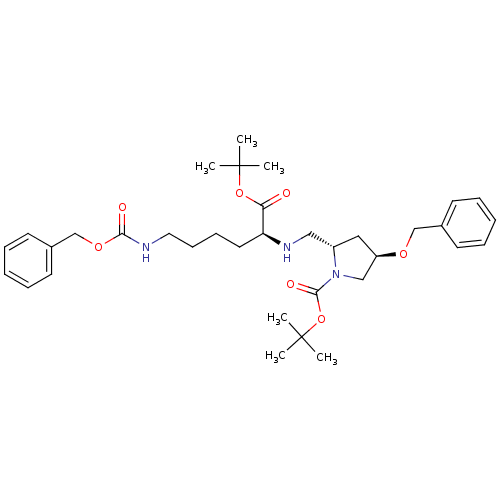
Affinity DataIC50: 220nMAssay Description:Inhibition of human MDR1 expressed in mouse NIH3T3 cells assessed as inhibition of drug efflux by flow cytometryMore data for this Ligand-Target Pair
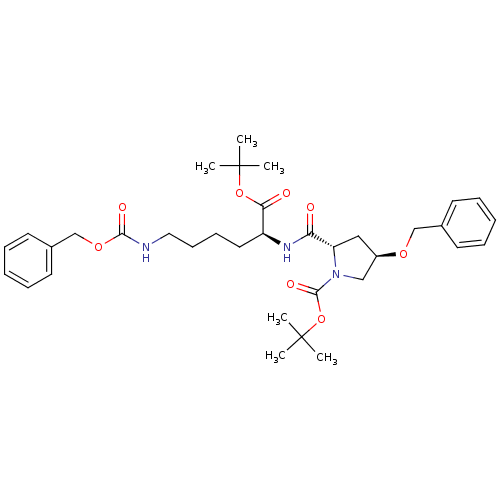
Affinity DataIC50: 730nMAssay Description:Inhibition of human MDR1 expressed in mouse NIH3T3 cells assessed as inhibition of drug efflux by flow cytometryMore data for this Ligand-Target Pair
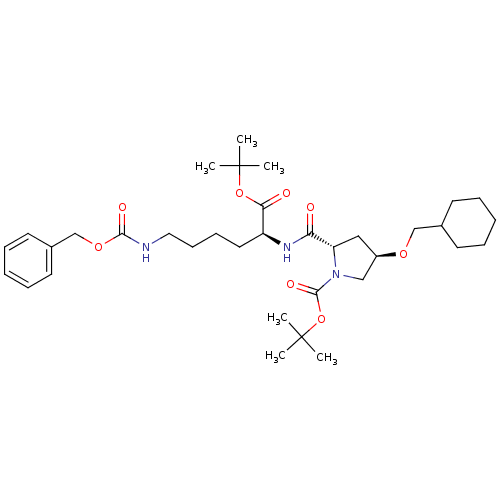
Affinity DataIC50: 1.13E+3nMAssay Description:Inhibition of human MDR1 expressed in mouse NIH3T3 cells assessed as inhibition of drug efflux by flow cytometryMore data for this Ligand-Target Pair
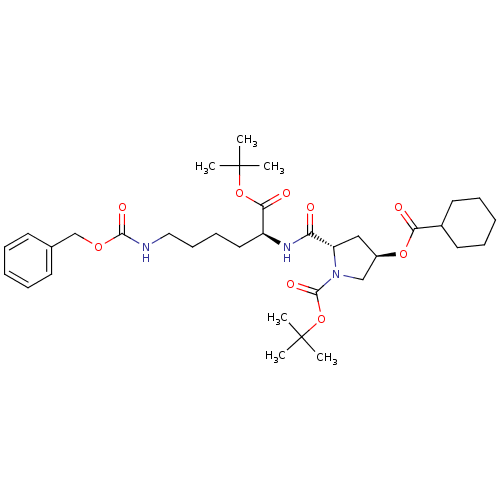
Affinity DataIC50: 1.41E+3nMAssay Description:Inhibition of human MDR1 expressed in mouse NIH3T3 cells assessed as inhibition of drug efflux by flow cytometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.55E+3nMAssay Description:Inhibition of human MDR1 expressed in mouse NIH3T3 cells assessed as inhibition of drug efflux by flow cytometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.68E+3nMAssay Description:Inhibition of human MDR1 expressed in mouse NIH3T3 cells assessed as inhibition of drug efflux by flow cytometryMore data for this Ligand-Target Pair