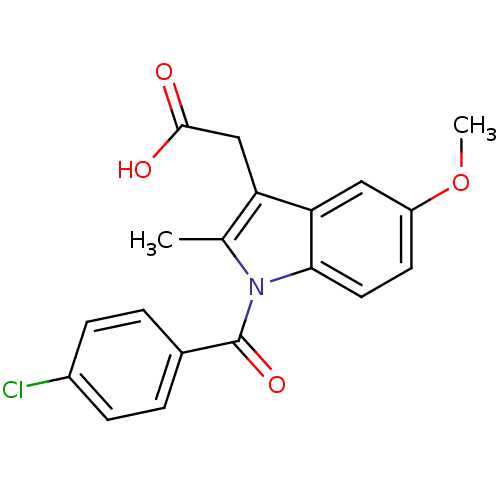
Affinity DataIC50: 120nMpH: 8.0 T: 2°CAssay Description:Reaction mixtures were prepared in 100 mM Tris�HCl buffer, pH 8.0 containing 1 �M heme and COX-1 or COX-2 and preincubated for 10 min in a waterbath ...More data for this Ligand-Target Pair
Affinity DataIC50: 600nMpH: 8.0 T: 2°CAssay Description:Reaction mixtures were prepared in 100 mM Tris�HCl buffer, pH 8.0 containing 1 �M heme and COX-1 or COX-2 and preincubated for 10 min in a waterbath ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10E+3nMpH: 8.0 T: 2°CAssay Description:Reaction mixtures were prepared in 100 mM Tris�HCl buffer, pH 8.0 containing 1 �M heme and COX-1 or COX-2 and preincubated for 10 min in a waterbath ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+3nMpH: 8.0 T: 2°CAssay Description:Reaction mixtures were prepared in 100 mM Tris�HCl buffer, pH 8.0 containing 1 �M heme and COX-1 or COX-2 and preincubated for 10 min in a waterbath ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.85E+4nMpH: 8.0 T: 2°CAssay Description:Reaction mixtures were prepared in 100 mM Tris�HCl buffer, pH 8.0 containing 1 �M heme and COX-1 or COX-2 and preincubated for 10 min in a waterbath ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMpH: 8.0 T: 2°CAssay Description:Reaction mixtures were prepared in 100 mM Tris�HCl buffer, pH 8.0 containing 1 �M heme and COX-1 or COX-2 and preincubated for 10 min in a waterbath ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMpH: 8.0 T: 2°CAssay Description:Reaction mixtures were prepared in 100 mM Tris�HCl buffer, pH 8.0 containing 1 �M heme and COX-1 or COX-2 and preincubated for 10 min in a waterbath ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMpH: 8.0 T: 2°CAssay Description:Reaction mixtures were prepared in 100 mM Tris�HCl buffer, pH 8.0 containing 1 �M heme and COX-1 or COX-2 and preincubated for 10 min in a waterbath ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMpH: 8.0 T: 2°CAssay Description:Reaction mixtures were prepared in 100 mM Tris�HCl buffer, pH 8.0 containing 1 �M heme and COX-1 or COX-2 and preincubated for 10 min in a waterbath ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMpH: 8.0 T: 2°CAssay Description:Reaction mixtures were prepared in 100 mM Tris�HCl buffer, pH 8.0 containing 1 �M heme and COX-1 or COX-2 and preincubated for 10 min in a waterbath ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMpH: 8.0 T: 2°CAssay Description:Reaction mixtures were prepared in 100 mM Tris�HCl buffer, pH 8.0 containing 1 �M heme and COX-1 or COX-2 and preincubated for 10 min in a waterbath ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMpH: 8.0 T: 2°CAssay Description:Reaction mixtures were prepared in 100 mM Tris�HCl buffer, pH 8.0 containing 1 �M heme and COX-1 or COX-2 and preincubated for 10 min in a waterbath ...More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMpH: 8.0 T: 2°CAssay Description:Reaction mixtures were prepared in 100 mM Tris�HCl buffer, pH 8.0 containing 1 �M heme and COX-1 or COX-2 and preincubated for 10 min in a waterbath ...More data for this Ligand-Target Pair
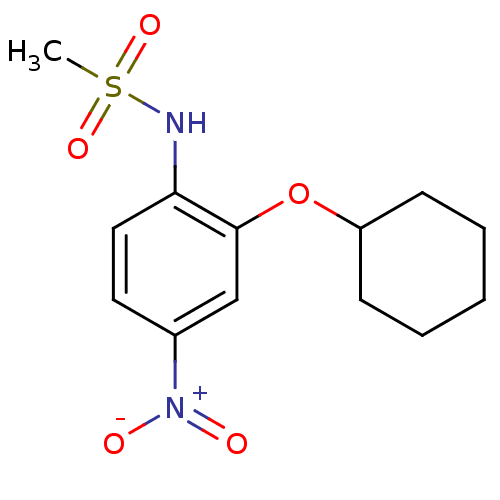
Affinity DataIC50: 2.13E+5nMpH: 8.0 T: 2°CAssay Description:Reaction mixtures were prepared in 100 mM Tris�HCl buffer, pH 8.0 containing 1 �M heme and COX-1 or COX-2 and preincubated for 10 min in a waterbath ...More data for this Ligand-Target Pair



 3D Structure (crystal)
3D Structure (crystal)

