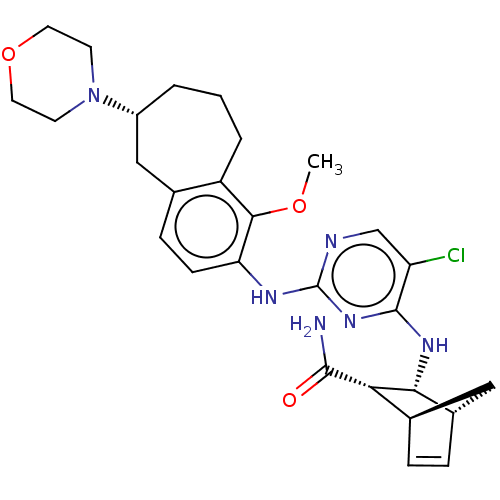
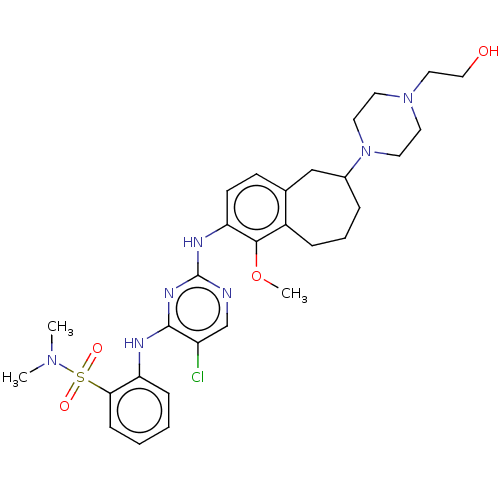
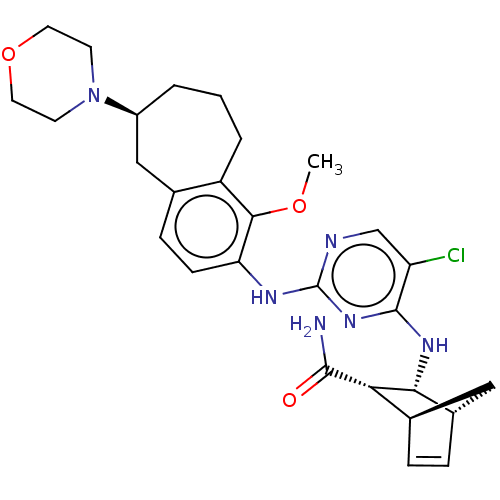
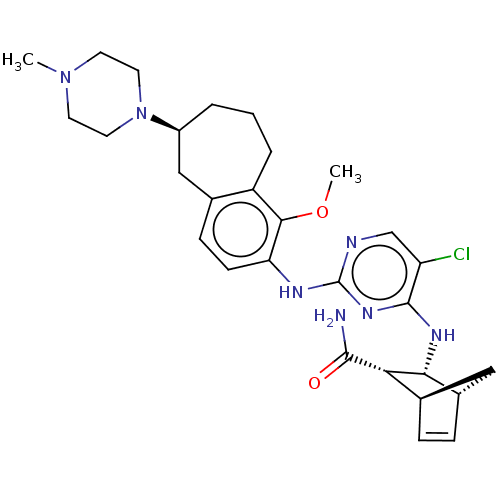
TargetFocal adhesion kinase 1(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 1nMAssay Description:Inhibition of human FAK expressed in baculovirus after 30 mins using biotinyl-amino-hexanoyl-EQEDEPEGDYFEWLE-amide as substrate by TRF assayMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 1.5nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 1.90nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 2nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetFocal adhesion kinase 1(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 2nMAssay Description:Inhibition of human FAK expressed in baculovirus after 30 mins using biotinyl-amino-hexanoyl-EQEDEPEGDYFEWLE-amide as substrate by TRF assayMore data for this Ligand-Target Pair
TargetFocal adhesion kinase 1(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 2.20nMAssay Description:Inhibition of human FAK expressed in baculovirus after 30 mins using biotinyl-amino-hexanoyl-EQEDEPEGDYFEWLE-amide as substrate by TRF assayMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 2.30nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 2.30nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetFocal adhesion kinase 1(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 2.40nMAssay Description:Inhibition of human FAK expressed in baculovirus after 30 mins using biotinyl-amino-hexanoyl-EQEDEPEGDYFEWLE-amide as substrate by TRF assayMore data for this Ligand-Target Pair
TargetFocal adhesion kinase 1(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 3.10nMAssay Description:Inhibition of human FAK expressed in baculovirus after 30 mins using biotinyl-amino-hexanoyl-EQEDEPEGDYFEWLE-amide as substrate by TRF assayMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 3.10nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 3.40nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetFocal adhesion kinase 1(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 3.80nMAssay Description:Inhibition of human FAK expressed in baculovirus after 30 mins using biotinyl-amino-hexanoyl-EQEDEPEGDYFEWLE-amide as substrate by TRF assayMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetFocal adhesion kinase 1(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:Inhibition of human FAK expressed in baculovirus after 30 mins using biotinyl-amino-hexanoyl-EQEDEPEGDYFEWLE-amide as substrate by TRF assayMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 5nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 5.70nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 5.80nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 6.10nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 6.30nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetFocal adhesion kinase 1(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 6.5nMAssay Description:Inhibition of human FAK expressed in baculovirus after 30 mins using biotinyl-amino-hexanoyl-EQEDEPEGDYFEWLE-amide as substrate by TRF assayMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 7nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 7nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetFocal adhesion kinase 1(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 7nMAssay Description:Inhibition of human FAK expressed in baculovirus after 30 mins using biotinyl-amino-hexanoyl-EQEDEPEGDYFEWLE-amide as substrate by TRF assayMore data for this Ligand-Target Pair
TargetFocal adhesion kinase 1(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 8.5nMAssay Description:Inhibition of human FAK expressed in baculovirus after 30 mins using biotinyl-amino-hexanoyl-EQEDEPEGDYFEWLE-amide as substrate by TRF assayMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 9nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 9nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetFocal adhesion kinase 1(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 9.30nMAssay Description:Inhibition of human FAK expressed in baculovirus after 30 mins using biotinyl-amino-hexanoyl-EQEDEPEGDYFEWLE-amide as substrate by TRF assayMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 18nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 20nMAssay Description:Inhibition of NPM-ALK phosphorylation in human SUP-M2 cells after 2 to 3 hrs by ELISAMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 20nMAssay Description:Inhibition of NPM-ALK phosphorylation in human SUP-M2 cells after 2 to 3 hrs by ELISAMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 22nMAssay Description:Inhibition of NPM-ALK phosphorylation in human SUP-M2 cells after 2 to 3 hrs by ELISAMore data for this Ligand-Target Pair
TargetFocal adhesion kinase 1(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 25nMAssay Description:Inhibition of human FAK expressed in baculovirus after 30 mins using biotinyl-amino-hexanoyl-EQEDEPEGDYFEWLE-amide as substrate by TRF assayMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 30nMAssay Description:Inhibition of NPM-ALK phosphorylation in human SUP-M2 cells after 2 to 3 hrs by ELISAMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 30nMAssay Description:Inhibition of NPM-ALK phosphorylation in human SUP-M2 cells after 2 to 3 hrs by ELISAMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 39nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 40nMAssay Description:Inhibition of NPM-ALK phosphorylation in human SUP-M2 cells after 2 to 3 hrs by ELISAMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 40nMAssay Description:Inhibition of NPM-ALK phosphorylation in human SUP-M2 cells after 2 to 3 hrs by ELISAMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 40nMAssay Description:Inhibition of NPM-ALK phosphorylation in human SUP-M2 cells after 2 to 3 hrs by ELISAMore data for this Ligand-Target Pair
TargetFocal adhesion kinase 1(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 42nMAssay Description:Inhibition of human FAK expressed in baculovirus after 30 mins using biotinyl-amino-hexanoyl-EQEDEPEGDYFEWLE-amide as substrate by TRF assayMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 44nMAssay Description:Inhibition of human ALK expressed in baculovirus using recombinant GST PLC-gamma as substrate assessed as phosphorylation of the substrate after 15 m...More data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 50nMAssay Description:Inhibition of NPM-ALK phosphorylation in human SUP-M2 cells after 2 to 3 hrs by ELISAMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 50nMAssay Description:Inhibition of NPM-ALK phosphorylation in human SUP-M2 cells after 2 to 3 hrs by ELISAMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 50nMAssay Description:Inhibition of NPM-ALK phosphorylation in human SUP-M2 cells after 2 to 3 hrs by ELISAMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 60nMAssay Description:Inhibition of NPM-ALK phosphorylation in human SUP-M2 cells after 2 to 3 hrs by ELISAMore data for this Ligand-Target Pair
TargetInsulin receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 65nMAssay Description:Inhibition of recombinant human INSR expressed in baculovirus using ATP as substrate after 15 mins by TRF assayMore data for this Ligand-Target Pair
TargetInsulin receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 71nMAssay Description:Inhibition of recombinant human INSR expressed in baculovirus using ATP as substrate after 15 mins by TRF assayMore data for this Ligand-Target Pair
TargetFocal adhesion kinase 1(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 80nMAssay Description:Inhibition of FAK phosphorylation in human HCC827 cells after 2 to 2.5 hrs by immunoblot analysisMore data for this Ligand-Target Pair
TargetInsulin receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 81nMAssay Description:Inhibition of recombinant human INSR expressed in baculovirus using ATP as substrate after 15 mins by TRF assayMore data for this Ligand-Target Pair
TargetALK tyrosine kinase receptor(Homo sapiens (Human))
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Teva Branded Pharmaceutical Products R & D
Curated by ChEMBL
Affinity DataIC50: 85nMAssay Description:Inhibition of EML4-ALK tyrosine phosphorylation in human NCI-H3122 cellsMore data for this Ligand-Target Pair