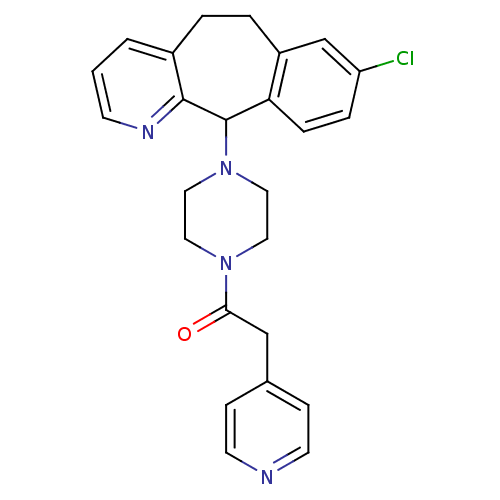
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 32nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
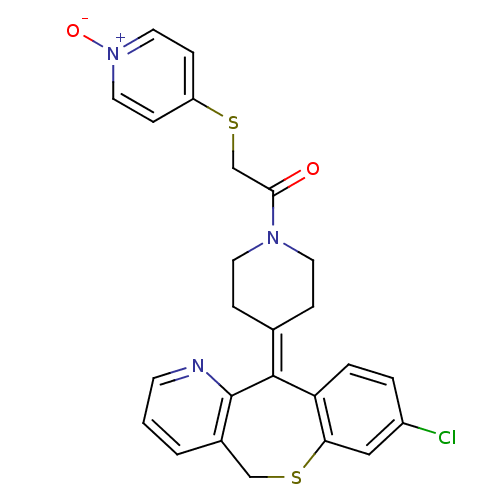
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 32nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 36nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 38nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 43nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 61nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 79nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 110nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 140nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 140nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 190nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 250nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 520nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 560nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 590nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 620nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 840nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 920nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.00E+3nMAssay Description:In vitro inhibition of Ras processing in COS cellsMore data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.00E+3nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.10E+3nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.12E+3nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.52E+3nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.70E+3nMAssay Description:In vitro inhibition of Ras processing in COS cellsMore data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.79E+3nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 2.10E+3nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 2.70E+3nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 3.40E+3nMAssay Description:In vitro FPT potency by measuring the ability to inhibit the transfer of [3H]-farnesyl from farnesyl pyrophosphate to H-Ras-CLVS.More data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 3.70E+3nMAssay Description:In vitro inhibition of Ras processing in COS cellsMore data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 3.80E+3nMAssay Description:In vitro inhibition of Ras processing in COS cellsMore data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 4.20E+3nMAssay Description:In vitro inhibition of Ras processing in COS cellsMore data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 5.00E+3nMAssay Description:In vitro inhibition of Ras processing in COS cellsMore data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:In vitro inhibition of Ras processing in COS cellsMore data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:In vitro inhibition of Ras processing in COS cellsMore data for this Ligand-Target Pair
TargetProtein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha(Homo sapiens (Human))
Schering-Plough Research Institute
Curated by ChEMBL
Schering-Plough Research Institute
Curated by ChEMBL
Affinity DataIC50: 2.00E+4nMAssay Description:In vitro inhibition of Ras processing in COS cellsMore data for this Ligand-Target Pair