Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
11-beta-hydroxysteroid dehydrogenase type 2
Ligand
BDBM13768
Substrate
BDBM13775
Meas. Tech.
Human and Mouse 11beta-HSD1 SPA Assay
IC50
20000±n/a nM
Citation
 Patel, JR; Shuai, Q; Dinges, J; Winn, M; Pliushchev, M; Fung, S; Monzon, K; Chiou, W; Wang, J; Pan, L; Wagaw, S; Engstrom, K; Kerdesky, FA; Longenecker, K; Judge, R; Qin, W; Imade, HM; Stolarik, D; Beno, DW; Brune, M; Chovan, LE; Sham, HL; Jacobson, P; Link, JT Discovery of adamantane ethers as inhibitors of 11beta-HSD-1: Synthesis and biological evaluation. Bioorg Med Chem Lett 17:750-5 (2007) [PubMed] Article
Patel, JR; Shuai, Q; Dinges, J; Winn, M; Pliushchev, M; Fung, S; Monzon, K; Chiou, W; Wang, J; Pan, L; Wagaw, S; Engstrom, K; Kerdesky, FA; Longenecker, K; Judge, R; Qin, W; Imade, HM; Stolarik, D; Beno, DW; Brune, M; Chovan, LE; Sham, HL; Jacobson, P; Link, JT Discovery of adamantane ethers as inhibitors of 11beta-HSD-1: Synthesis and biological evaluation. Bioorg Med Chem Lett 17:750-5 (2007) [PubMed] Article More Info.:
Target
Name:
11-beta-hydroxysteroid dehydrogenase type 2
Synonyms:
11-DH2 | 11-beta-HSD2 | 11-beta-Hydroxysteroid Dehydrogenase 2 (11-beta-HSD2) | 11-beta-hydroxysteroid dehydrogenase | 11-beta-hydroxysteroid dehydrogenase 2 | 11-beta-hydroxysteroid dehydrogenase type 2 | 11-beta-hydroxysteroid dehydrogenase type 2 (11-beta-HSD2) | Corticosteroid 11-beta-dehydrogenase isozyme 2 | DHI2_HUMAN | HSD11B2 | HSD11K | NAD-dependent 11-beta-hydroxysteroid dehydrogenase | SDR9C3
Type:
Enzyme
Mol. Mass.:
44141.72
Organism:
Homo sapiens (Human)
Description:
Purified recombinant human 11beta-HSD2.
Residue:
405
Sequence:
MERWPWPSGGAWLLVAARALLQLLRSDLRLGRPLLAALALLAALDWLCQRLLPPPAALAVLAAAGWIALSRLARPQRLPVATRAVLITGCDSGFGKETAKKLDSMGFTVLATVLELNSPGAIELRTCCSPRLRLLQMDLTKPGDISRVLEFTKAHTTSTGLWGLVNNAGHNEVVADAELSPVATFRSCMEVNFFGALELTKGLLPLLRSSRGRIVTVGSPAGDMPYPCLGAYGTSKAAVALLMDTFSCELLPWGVKVSIIQPGCFKTESVRNVGQWEKRKQLLLANLPQELLQAYGKDYIEHLHGQFLHSLRLAMSDLTPVVDAITDALLAARPRRRYYPGQGLGLMYFIHYYLPEGLRRRFLQAFFISHCLPRALQPGQPGTTPPQDAAQDPNLSPGPSPAVAR
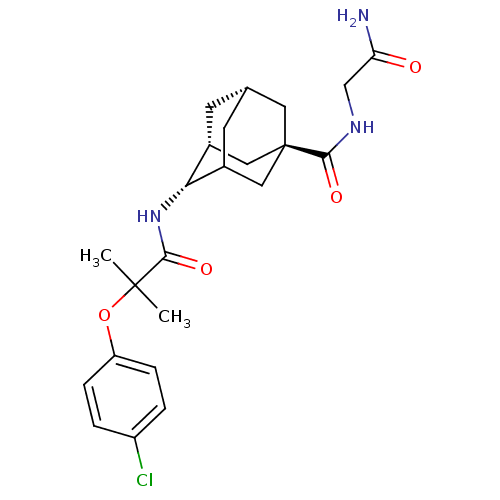
Inhibitor
Name:
BDBM13768
Synonyms:
N-[(1R,2S,5R,7S)-5-[(carbamoylmethyl)carbamoyl]adamantan-2-yl]-2-(4-chlorophenoxy)-2-methylpropanamide | adamantane ether 22
Type:
Small organic molecule
Emp. Form.:
C23H30ClN3O4
Mol. Mass.:
447.955
SMILES:
CC(C)(Oc1ccc(Cl)cc1)C(=O)N[C@H]1C2C[C@@H]3C[C@@H]1C[C@](C3)(C2)C(=O)NCC(N)=O |r,wU:17.17,19.19,14.14,wD:21.27,TLB:23:21:18:16.15.14,THB:16:17:20:23.15.14,22:17:14:23.20.21,24:21:18:16.15.14,(10.84,-3.12,;11.61,-4.45,;12.7,-3.37,;12.94,-5.22,;14.43,-4.83,;14.43,-3.29,;15.76,-2.52,;17.1,-3.29,;18.43,-2.52,;17.1,-4.83,;15.76,-5.6,;10.27,-5.22,;10.27,-6.76,;8.94,-4.45,;7.61,-5.22,;7.61,-6.76,;6.94,-7.93,;4.72,-7.16,;4.72,-5.12,;6.04,-4.32,;4.24,-5.07,;4.24,-6.31,;3.21,-7.32,;6.26,-7.03,;2.91,-5.54,;2.51,-4.05,;1.58,-6.31,;1.58,-7.85,;.24,-8.62,;-1.09,-7.85,;.24,-10.16,)|
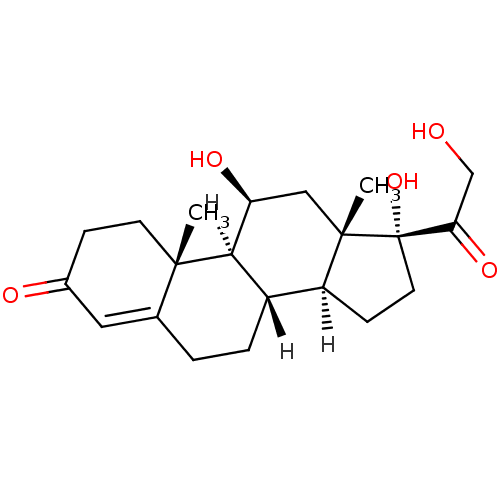
Substrate
Name:
BDBM13775
Synonyms:
(1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-hydroxyacetyl)-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-6-en-5-one | 11beta,17alpha,21-Trihydroxy-4-pregnene-3,20-dione | 3H-cortisol | HYDROCORTISONE | US10188667, Example 00023 | [3H]cortisol | cortisol
Type:
Steroid
Emp. Form.:
C21H30O5
Mol. Mass.:
362.4599
SMILES:
[H][C@@]12CC[C@](O)(C(=O)CO)[C@@]1(C)C[C@H](O)[C@@]1([H])[C@@]2([H])CCC2=CC(=O)CC[C@]12C |t:23|