Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Glutaminase kidney isoform, mitochondrial
Ligand
BDBM50503314
Substrate
n/a
Meas. Tech.
ChEMBL_1809963 (CHEMBL4309423)
IC50
2000±n/a nM
Citation
 Zimmermann, SC; Duvall, B; Tsukamoto, T Recent Progress in the Discovery of Allosteric Inhibitors of Kidney-Type Glutaminase. J Med Chem 62:46-59 (2019) [PubMed] Article
Zimmermann, SC; Duvall, B; Tsukamoto, T Recent Progress in the Discovery of Allosteric Inhibitors of Kidney-Type Glutaminase. J Med Chem 62:46-59 (2019) [PubMed] Article More Info.:
Target
Name:
Glutaminase kidney isoform, mitochondrial
Synonyms:
GLS | GLS1 | GLSK_HUMAN | Glutaminase 1 | K-glutaminase | KIAA0838 | L-glutamine amidohydrolase
Type:
Protein
Mol. Mass.:
73471.89
Organism:
Homo sapiens (Human)
Description:
O94925
Residue:
669
Sequence:
MMRLRGSGMLRDLLLRSPAGVSATLRRAQPLVTLCRRPRGGGRPAAGPAAAARLHPWWGGGGWPAEPLARGLSSSPSEILQELGKGSTHPQPGVSPPAAPAAPGPKDGPGETDAFGNSEGKELVASGENKIKQGLLPSLEDLLFYTIAEGQEKIPVHKFITALKSTGLRTSDPRLKECMDMLRLTLQTTSDGVMLDKDLFKKCVQSNIVLLTQAFRRKFVIPDFMSFTSHIDELYESAKKQSGGKVADYIPQLAKFSPDLWGVSVCTVDGQRHSTGDTKVPFCLQSCVKPLKYAIAVNDLGTEYVHRYVGKEPSGLRFNKLFLNEDDKPHNPMVNAGAIVVTSLIKQGVNNAEKFDYVMQFLNKMAGNEYVGFSNATFQSERESGDRNFAIGYYLKEKKCFPEGTDMVGILDFYFQLCSIEVTCESASVMAATLANGGFCPITGERVLSPEAVRNTLSLMHSCGMYDFSGQFAFHVGLPAKSGVAGGILLVVPNVMGMMCWSPPLDKMGNSVKGIHFCHDLVSLCNFHNYDNLRHFAKKLDPRREGGDQRVKSVINLLFAAYTGDVSALRRFALSAMDMEQRDYDSRTALHVAAAEGHVEVVKFLLEACKVNPFPKDRWNNTPMDEALHFGHHDVFKILQEYQVQYTPQGDSDNGKENQTVHKNLDGLL
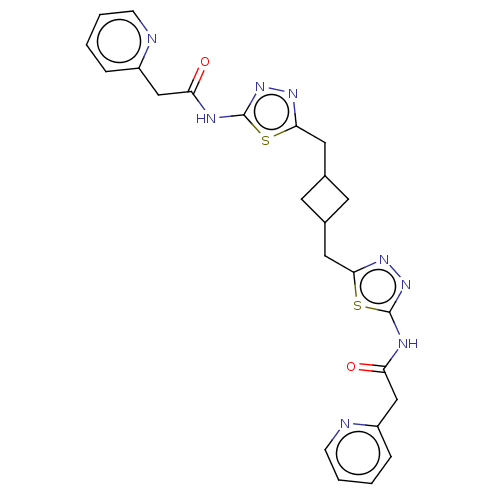
Inhibitor
Name:
BDBM50503314
Synonyms:
CHEMBL4457558
Type:
Small organic molecule
Emp. Form.:
C24H24N8O2S2
Mol. Mass.:
520.63
SMILES:
O=C(Cc1ccccn1)Nc1nnc(CC2CC(Cc3nnc(NC(=O)Cc4ccccn4)s3)C2)s1 |(80.99,-35.17,;81,-36.71,;82.33,-37.48,;83.66,-36.7,;83.66,-35.17,;84.97,-34.4,;86.32,-35.17,;86.32,-36.71,;84.99,-37.48,;79.67,-37.48,;78.34,-36.72,;78.81,-35.25,;77.57,-34.35,;76.33,-35.25,;75,-34.48,;73.66,-35.24,;73.27,-36.73,;71.78,-36.33,;70.45,-37.1,;69.12,-36.33,;68.64,-34.86,;67.11,-34.86,;66.63,-36.33,;65.29,-37.09,;63.96,-36.31,;63.97,-34.77,;62.62,-37.07,;61.29,-36.29,;61.31,-34.75,;60,-33.98,;58.65,-34.73,;58.65,-36.28,;59.98,-37.05,;67.88,-37.23,;72.17,-34.84,;76.8,-36.72,)|