Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Leukotriene A-4 hydrolase
Ligand
BDBM50266316
Substrate
n/a
Meas. Tech.
ChEMBL_544515 (CHEMBL1013487)
IC50
55±n/a nM
Citation
 Enomoto, H; Morikawa, Y; Miyake, Y; Tsuji, F; Mizuchi, M; Suhara, H; Fujimura, K; Horiuchi, M; Ban, M Synthesis and biological evaluation of N-mercaptoacylcysteine derivatives as leukotriene A4 hydrolase inhibitors. Bioorg Med Chem Lett 19:442-6 (2008) [PubMed] Article
Enomoto, H; Morikawa, Y; Miyake, Y; Tsuji, F; Mizuchi, M; Suhara, H; Fujimura, K; Horiuchi, M; Ban, M Synthesis and biological evaluation of N-mercaptoacylcysteine derivatives as leukotriene A4 hydrolase inhibitors. Bioorg Med Chem Lett 19:442-6 (2008) [PubMed] Article More Info.:
Target
Name:
Leukotriene A-4 hydrolase
Synonyms:
LKHA4_CAVPO | LTA4H
Type:
PROTEIN
Mol. Mass.:
68972.78
Organism:
Cavia porcellus
Description:
ChEMBL_544515
Residue:
611
Sequence:
MPEVVDTCSLASPATVCRTKHLHLRCSVDFTRRALTGVAALTIQSQEDNLRSLILDTKDLTIEKVVINGQEVKYALGEKQSYKGSPMEISLPIALSKNQEVVIEISFETSPKSSALQWLTPEQTSGKEHPYLFSQCQAIHCRAFLPCQDTPSVKLTYTAEVSVPKELVALMSAIRDGEAPDPADPSRKIYKFSQKVPIPCYLIALVVGALESRKIGPRTLVWSEKEQVDKSAYEFSETESMLKIAEDLGGPYVWGQYDRLVLPPSFSYGGMENPCLTFVTPTLLAGDKSLSNVIAHEISHTWTGNLVTNKTWDHFWLNEGHTVYLERHICGRLFGEKFRHFHALGGWGELQNTVKTLGETQAFTKLVVDLTDTDPDVAYSSVPYEKGFALLFHLEQLLGGPEVFLGFLKAYVEKFSYKSITTDDWKNFLFSHFKDKVDILNQVDWDAWLYSPGLPPIKPNYDMTLTNACIALSQRWITAKEKDLNTFSATDLKDLSSHQVNEFLAQVLQRAPLPLGHVKRMQEVYNCNAINNSEIRFRWLRLCIQSKWEEAIPLALKMATEQGRMKFTRPLFKDLAAFDKSHDQAIQTYHAHKASMHPVTAMLVGKDLKVE
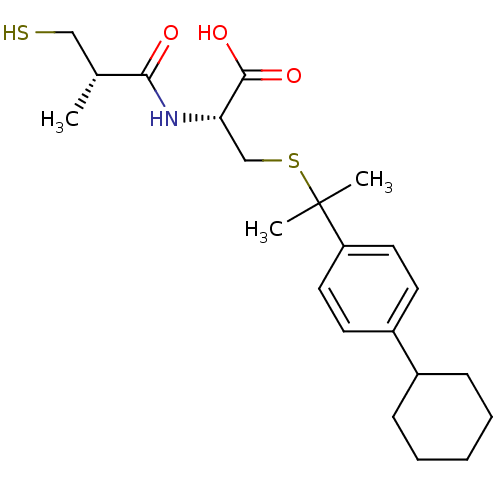
Inhibitor
Name:
BDBM50266316
Synonyms:
(R)-3-(2-(4-cyclohexylphenyl)propan-2-ylthio)-2-((S)-3-mercapto-2-methylpropanamido)propanoic acid | CHEMBL456045
Type:
Small organic molecule
Emp. Form.:
C22H33NO3S2
Mol. Mass.:
423.632
SMILES:
C[C@H](CS)C(=O)N[C@@H](CSC(C)(C)c1ccc(cc1)C1CCCCC1)C(O)=O |r|