Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Prostaglandin G/H synthase 1
Ligand
BDBM17638
Substrate
n/a
Meas. Tech.
ChEBML_159270
IC50
6±n/a nM
Citation
 Leblanc, Y; Black, WC; Chan, CC; Charleson, S; Delorme, D; Denis, D; Gauthier, JY; Grimm, EL; Gordon, R; Guay, D; Hamel, P; Kargman, S; Lau, CK; Mancini, J; Ouellet, M; Percival, D; Roy, P; Skorey, K; Tagari, P; Vickers, P Synthesis and biological evaluation of both enantiomers of L-761,000 as inhibitors of cyclooxygenase 1 and 2 Bioorg Med Chem Lett 6:731-736 (1996) Article
Leblanc, Y; Black, WC; Chan, CC; Charleson, S; Delorme, D; Denis, D; Gauthier, JY; Grimm, EL; Gordon, R; Guay, D; Hamel, P; Kargman, S; Lau, CK; Mancini, J; Ouellet, M; Percival, D; Roy, P; Skorey, K; Tagari, P; Vickers, P Synthesis and biological evaluation of both enantiomers of L-761,000 as inhibitors of cyclooxygenase 1 and 2 Bioorg Med Chem Lett 6:731-736 (1996) Article More Info.:
Target
Name:
Prostaglandin G/H synthase 1
Synonyms:
Cox-1 | Cox1 | Cyclooxygenase | Cyclooxygenase-1 | PGH1_RAT | Prostaglandin G/H synthase (cyclooxygenase) | Ptgs1
Type:
Enzyme Catalytic Domain
Mol. Mass.:
69033.56
Organism:
RAT
Description:
COX-1 0 RAT::Q63921
Residue:
602
Sequence:
MSRRSLSLQFPLLLLLLLLPPPPVLLTDAGVPSPVNPCCYYPCQNQGVCVRFGLDHYQCDCTRTGYSGPNCTIPEIWTWLRSSLRPSPSFTHFLLTHGYWIWEFVNATFIREVLMRLVITVRSNLIPSPPTYNTAHDYISWESFSNVSYYTRILPSVPKDCPTPMGTKGKKQLPDIHLLAQRLLLRREFIPGPQGTNVLFAFFAQHFTHQFFKTSGKMGPGFTKALGHGVDLGHIYGDSLERQYHLRLFKDGKLKYQVLDGEVYPPSVEQASVLMRYPPGVPPEKQMAVGQEVFGLLPGLMLFSTIWLREHNRVCDLLKEEHPTWDDEQLFQTTRLILIGETIKIIIEEYVQHLSGYFLQLKFDPELLFRAQFQYRNRIALEFNHLYHWHPLMPDSFQVGSQEYSYEQFLFNTSMLVDYGVEALVDAFSRQRAGRIGGGRNFDYHVLHVAEDVIKESREMRLQSFNEYRKRFGLKPYTSFQEFTGEKEMAAELEELYGDIDALEFYPGLMLEKCQPNSLFGESMIEMGAPFSLKGLLGNPICSPEYWKPSTFGGDVGFNIVNTASLKKLVCLNTKTCPYVSFRVPDYPGDDGSVFVRPSTEL
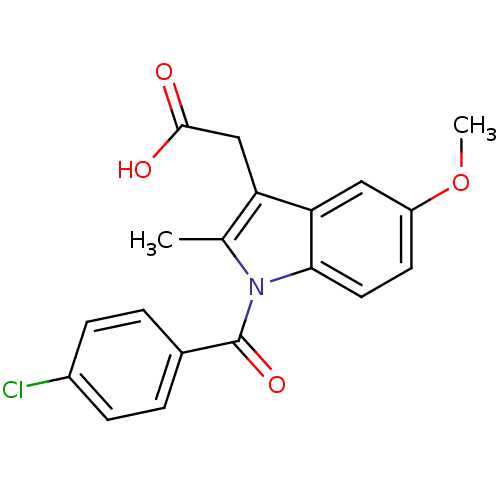
Inhibitor
Name:
BDBM17638
Synonyms:
2-{1-[(4-chlorophenyl)carbonyl]-5-methoxy-2-methyl-1H-indol-3-yl}acetic acid | CHEMBL6 | Indocin | Indomethacin | US11478464, Compound Indomethacin | US11786535, Compound Indomethacin | US9271961, Indomethacin | indometacin
Type:
Small organic molecule
Emp. Form.:
C19H16ClNO4
Mol. Mass.:
357.788
SMILES:
COc1ccc2n(C(=O)c3ccc(Cl)cc3)c(C)c(CC(O)=O)c2c1