Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 2D6
Ligand
BDBM50333708
Substrate
n/a
Meas. Tech.
ChEMBL_700453 (CHEMBL1647992)
IC50
>10000±n/a nM
Citation
 Tyagarajan, S; Chakravarty, PK; Park, M; Zhou, B; Herrington, JB; Ratliff, K; Bugianesi, RM; Williams, B; Haedo, RJ; Swensen, AM; Warren, VA; Smith, M; Garcia, M; Kaczorowski, GJ; McManus, OB; Lyons, KA; Li, X; Madeira, M; Karanam, B; Green, M; Forrest, MJ; Abbadie, C; McGowan, E; Mistry, S; Jochnowitz, N; Duffy, JL A potent and selective indole N-type calcium channel (Ca(v)2.2) blocker for the treatment of pain. Bioorg Med Chem Lett 21:869-73 (2011) [PubMed] Article
Tyagarajan, S; Chakravarty, PK; Park, M; Zhou, B; Herrington, JB; Ratliff, K; Bugianesi, RM; Williams, B; Haedo, RJ; Swensen, AM; Warren, VA; Smith, M; Garcia, M; Kaczorowski, GJ; McManus, OB; Lyons, KA; Li, X; Madeira, M; Karanam, B; Green, M; Forrest, MJ; Abbadie, C; McGowan, E; Mistry, S; Jochnowitz, N; Duffy, JL A potent and selective indole N-type calcium channel (Ca(v)2.2) blocker for the treatment of pain. Bioorg Med Chem Lett 21:869-73 (2011) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 2D6
Synonyms:
CP2D6_HUMAN | CYP2D6 | CYP2DL1 | CYPIID6 | Cytochrome P450 2D6 (CYP2D6) | Debrisoquine 4-hydroxylase | P450-DB1
Type:
Protein
Mol. Mass.:
55774.82
Organism:
Homo sapiens (Human)
Description:
P10635
Residue:
497
Sequence:
MGLEALVPLAVIVAIFLLLVDLMHRRQRWAARYPPGPLPLPGLGNLLHVDFQNTPYCFDQLRRRFGDVFSLQLAWTPVVVLNGLAAVREALVTHGEDTADRPPVPITQILGFGPRSQGVFLARYGPAWREQRRFSVSTLRNLGLGKKSLEQWVTEEAACLCAAFANHSGRPFRPNGLLDKAVSNVIASLTCGRRFEYDDPRFLRLLDLAQEGLKEESGFLREVLNAVPVLLHIPALAGKVLRFQKAFLTQLDELLTEHRMTWDPAQPPRDLTEAFLAEMEKAKGNPESSFNDENLRIVVADLFSAGMVTTSTTLAWGLLLMILHPDVQRRVQQEIDDVIGQVRRPEMGDQAHMPYTTAVIHEVQRFGDIVPLGVTHMTSRDIEVQGFRIPKGTTLITNLSSVLKDEAVWEKPFRFHPEHFLDAQGHFVKPEAFLPFSAGRRACLGEPLARMELFLFFTSLLQHFSFSVPTGQPRPSHHGVFAFLVSPSPYELCAVPR
Inhibitor
Name:
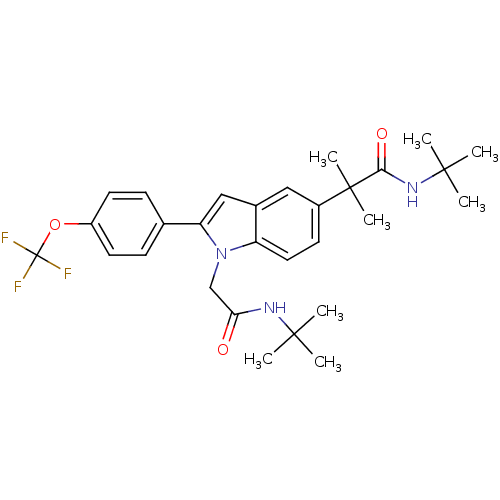
BDBM50333708
Synonyms:
CHEMBL1643735 | N-tert-butyl-2-(1-(2-(tert-butylamino)-2-oxoethyl)-2-(4-(trifluoromethoxy)phenyl)-1H-indol-5-yl)-2-methylpropanamide
Type:
Small organic molecule
Emp. Form.:
C29H36F3N3O3
Mol. Mass.:
531.6096
SMILES:
CC(C)(C)NC(=O)Cn1c(cc2cc(ccc12)C(C)(C)C(=O)NC(C)(C)C)-c1ccc(OC(F)(F)F)cc1