Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Mitogen-activated protein kinase 14
Ligand
BDBM16016
Substrate
n/a
Meas. Tech.
ChEMBL_701209 (CHEMBL1648829)
IC50
4±n/a nM
Citation
 Probst, GD; Bowers, S; Sealy, JM; Truong, AP; Hom, RK; Galemmo, RA; Konradi, AW; Sham, HL; Quincy, DA; Pan, H; Yao, N; Lin, M; Tóth, G; Artis, DR; Zmolek, W; Wong, K; Qin, A; Lorentzen, C; Nakamura, DF; Quinn, KP; Sauer, JM; Powell, K; Ruslim, L; Wright, S; Chereau, D; Ren, Z; Anderson, JP; Bard, F; Yednock, TA; Griswold-Prenner, I Highly selective c-Jun N-terminal kinase (JNK) 2 and 3 inhibitors with in vitro CNS-like pharmacokinetic properties prevent neurodegeneration. Bioorg Med Chem Lett 21:315-9 (2010) [PubMed] Article
Probst, GD; Bowers, S; Sealy, JM; Truong, AP; Hom, RK; Galemmo, RA; Konradi, AW; Sham, HL; Quincy, DA; Pan, H; Yao, N; Lin, M; Tóth, G; Artis, DR; Zmolek, W; Wong, K; Qin, A; Lorentzen, C; Nakamura, DF; Quinn, KP; Sauer, JM; Powell, K; Ruslim, L; Wright, S; Chereau, D; Ren, Z; Anderson, JP; Bard, F; Yednock, TA; Griswold-Prenner, I Highly selective c-Jun N-terminal kinase (JNK) 2 and 3 inhibitors with in vitro CNS-like pharmacokinetic properties prevent neurodegeneration. Bioorg Med Chem Lett 21:315-9 (2010) [PubMed] Article More Info.:
Target
Name:
Mitogen-activated protein kinase 14
Synonyms:
CSAID-binding protein | CSBP | CSBP1 | CSBP2 | CSPB1 | Cytokine suppressive anti-inflammatory drug-binding protein | MAP kinase 14 | MAP kinase MXI2 | MAP kinase p38 alpha | MAPK 14 | MAPK14 | MAX-interacting protein 2 | MK14_HUMAN | MXI2 | Mitogen-activated protein kinase p38 alpha | SAPK2A | Stress-activated protein kinase 2a | p38 MAP kinase alpha/beta
Type:
Serine/threonine-protein kinase
Mol. Mass.:
41286.76
Organism:
Homo sapiens (Human)
Description:
Q16539
Residue:
360
Sequence:
MSQERPTFYRQELNKTIWEVPERYQNLSPVGSGAYGSVCAAFDTKTGLRVAVKKLSRPFQSIIHAKRTYRELRLLKHMKHENVIGLLDVFTPARSLEEFNDVYLVTHLMGADLNNIVKCQKLTDDHVQFLIYQILRGLKYIHSADIIHRDLKPSNLAVNEDCELKILDFGLARHTDDEMTGYVATRWYRAPEIMLNWMHYNQTVDIWSVGCIMAELLTGRTLFPGTDHIDQLKLILRLVGTPGAELLKKISSESARNYIQSLTQMPKMNFANVFIGANPLAVDLLEKMLVLDSDKRITAAQALAHAYFAQYHDPDDEPVADPYDQSFESRDLLIDEWKSLTYDEVISFVPPPLDQEEMES
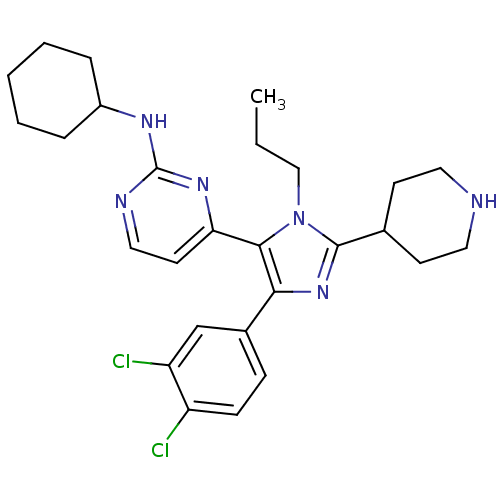
Inhibitor
Name:
BDBM16016
Synonyms:
CHEMBL437747 | N-cyclohexyl-4-[4-(3,4-dichlorophenyl)-2-(piperidin-4-yl)-1-propyl-1H-imidazol-5-yl]pyrimidin-2-amine | N-cyclohexyl-4-[4-(3,4-dichlorophenyl)-2-piperidin-4-yl-1-propyl-1H-imidazol-5-yl]pyrimidin-2-amine | imidazole-pyrimidine compound 2
Type:
Small organic molecule
Emp. Form.:
C27H34Cl2N6
Mol. Mass.:
513.505
SMILES:
CCCn1c(nc(c1-c1ccnc(NC2CCCCC2)n1)-c1ccc(Cl)c(Cl)c1)C1CCNCC1