Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Glutamate receptor ionotropic, NMDA 2B
Ligand
BDBM50032651
Substrate
n/a
Meas. Tech.
ChEMBL_748229 (CHEMBL1781192)
Ki
4.6±n/a nM
Citation
 Brown, DG; Maier, DL; Sylvester, MA; Hoerter, TN; Menhaji-Klotz, E; Lasota, CC; Hirata, LT; Wilkins, DE; Scott, CW; Trivedi, S; Chen, T; McCarthy, DJ; Maciag, CM; Sutton, EJ; Cumberledge, J; Mathisen, D; Roberts, J; Gupta, A; Liu, F; Elmore, CS; Alhambra, C; Krumrine, JR; Wang, X; Ciaccio, PJ; Wood, MW; Campbell, JB; Johansson, MJ; Xia, J; Wen, X; Jiang, J; Wang, X; Peng, Z; Hu, T; Wang, J 2,6-Disubstituted pyrazines and related analogs as NR2B site antagonists of the NMDA receptor with anti-depressant activity. Bioorg Med Chem Lett 21:3399-403 (2011) [PubMed] Article
Brown, DG; Maier, DL; Sylvester, MA; Hoerter, TN; Menhaji-Klotz, E; Lasota, CC; Hirata, LT; Wilkins, DE; Scott, CW; Trivedi, S; Chen, T; McCarthy, DJ; Maciag, CM; Sutton, EJ; Cumberledge, J; Mathisen, D; Roberts, J; Gupta, A; Liu, F; Elmore, CS; Alhambra, C; Krumrine, JR; Wang, X; Ciaccio, PJ; Wood, MW; Campbell, JB; Johansson, MJ; Xia, J; Wen, X; Jiang, J; Wang, X; Peng, Z; Hu, T; Wang, J 2,6-Disubstituted pyrazines and related analogs as NR2B site antagonists of the NMDA receptor with anti-depressant activity. Bioorg Med Chem Lett 21:3399-403 (2011) [PubMed] Article More Info.:
Target
Name:
Glutamate receptor ionotropic, NMDA 2B
Synonyms:
GluN2B | Glutamate [NMDA] receptor subunit epsilon 2 | Grin2b | N-methyl D-aspartate receptor subtype 2B | NMDA receptor subunit N2B (GluN2B) | NMDAR2B | NMDE2_RAT | NR2B
Type:
Protein
Mol. Mass.:
166077.66
Organism:
Rattus norvegicus (Rat)
Description:
Q00960
Residue:
1482
Sequence:
MKPSAECCSPKFWLVLAVLAVSGSKARSQKSPPSIGIAVILVGTSDEVAIKDAHEKDDFHHLSVVPRVELVAMNETDPKSIITRICDLMSDRKIQGVVFADDTDQEAIAQILDFISAQTLTPILGIHGGSSMIMADKDESSMFFQFGPSIEQQASVMLNIMEEYDWYIFSIVTTYFPGYQDFVNKIRSTIENSFVGWELEEVLLLDMSLDDGDSKIQNQLKKLQSPIILLYCTKEEATYIFEVANSVGLTGYGYTWIVPSLVAGDTDTVPSEFPTGLISVSYDEWDYGLPARVRDGIAIITTAASDMLSEHSFIPEPKSSCYNTHEKRIYQSNMLNRYLINVTFEGRNLSFSEDGYQMHPKLVIILLNKERKWERVGKWKDKSLQMKYYVWPRMCPETEEQEDDHLSIVTLEEAPFVIVESVDPLSGTCMRNTVPCQKRIISENKTDEEPGYIKKCCKGFCIDILKKISKSVKFTYDLYLVTNGKHGKKINGTWNGMIGEVVMKRAYMAVGSLTINEERSEVVDFSVPFIETGISVMVSRSNGTVSPSAFLEPFSADVWVMMFVMLLIVSAVAVFVFEYFSPVGYNRCLADGREPGGPSFTIGKAIWLLWGLVFNNSVPVQNPKGTTSKIMVSVWAFFAVIFLASYTANLAAFMIQEEYVDQVSGLSDKKFQRPNDFSPPFRFGTVPNGSTERNIRNNYAEMHAYMGKFNQRGVDDALLSLKTGKLDAFIYDAAVLNYMAGRDEGCKLVTIGSGKVFASTGYGIAIQKDSGWKRQVDLAILQLFGDGEMEELEALWLTGICHNEKNEVMSSQLDIDNMAGVFYMLGAAMALSLITFICEHLFYWQFRHCFMGVCSGKPGMVFSISRGIYSCIHGVAIEERQSVMNSPTATMNNTHSNILRLLRTAKNMANLSGVNGSPQSALDFIRRESSVYDISEHRRSFTHSDCKSYNNPPCEENLFSDYISEVERTFGNLQLKDSNVYQDHYHHHHRPHSIGSTSSIDGLYDCDNPPFTTQPRSISKKPLDIGLPSSKHSQLSDLYGKFSFKSDRYSGHDDLIRSDVSDISTHTVTYGNIEGNAAKRRKQQYKDSLKKRPASAKSRREFDEIELAYRRRPPRSPDHKRYFRDKEGLRDFYLDQFRTKENSPHWEHVDLTDIYKERSDDFKRDSVSGGGPCTNRSHLKHGTGEKHGVVGGVPAPWEKNLTNVDWEDRSGGNFCRSCPSKLHNYSSTVAGQNSGRQACIRCEACKKAGNLYDISEDNSLQELDQPAAPVAVTSNASSTKYPQSPTNSKAQKKNRNKLRRQHSYDTFVDLQKEEAALAPRSVSLKDKGRFMDGSPYAHMFEMPAGESSFANKSSVPTAGHHHNNPGSGYMLSKSLYPDRVTQNPFIPTFGDDQCLLHGSKSYFFRQPTVAGASKTRPDFRALVTNKPVVVTLHGAVPGRFQKDICIGNQSNPCVPNNKNPRAFNGSSNGHVYEKLSSIESDV
Inhibitor
Name:
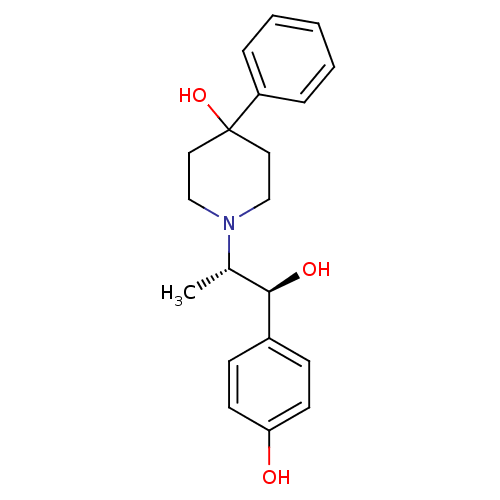
BDBM50032651
Synonyms:
1-((1S,2S)-1-hydroxy-1-(4-hydroxyphenyl)propan-2-yl)-4-phenylpiperidin-4-ol | 1-[(1S,2S)-2-Hydroxy-2-(4-hydroxy-phenyl)-1-methyl-ethyl]-4-phenyl-piperidin-4-ol | 1-[(S)-2-(S)-Hydroxy-2-(4-hydroxy-phenyl)-1-methyl-ethyl]-4-phenyl-piperidin-4-ol | CHEMBL17350 | CP-101,606 | CP-101606 | TRAXOPRODIL
Type:
Small organic molecule
Emp. Form.:
C20H25NO3
Mol. Mass.:
327.4174
SMILES:
C[C@@H]([C@@H](O)c1ccc(O)cc1)N1CCC(O)(CC1)c1ccccc1