Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Prostaglandin G/H synthase 2
Ligand
BDBM17638
Substrate
n/a
Meas. Tech.
ChEMBL_862693 (CHEMBL2174241)
IC50
20000±n/a nM
Citation
 Arisawa, M; Kasaya, Y; Obata, T; Sasaki, T; Nakamura, T; Araki, T; Yamamoto, K; Sasaki, A; Yamano, A; Ito, M; Abe, H; Ito, Y; Shuto, S Design and synthesis of indomethacin analogues that inhibit P-glycoprotein and/or multidrug resistant protein without COX inhibitory activity. J Med Chem 55:8152-63 (2012) [PubMed] Article
Arisawa, M; Kasaya, Y; Obata, T; Sasaki, T; Nakamura, T; Araki, T; Yamamoto, K; Sasaki, A; Yamano, A; Ito, M; Abe, H; Ito, Y; Shuto, S Design and synthesis of indomethacin analogues that inhibit P-glycoprotein and/or multidrug resistant protein without COX inhibitory activity. J Med Chem 55:8152-63 (2012) [PubMed] Article More Info.:
Target
Name:
Prostaglandin G/H synthase 2
Synonyms:
COX2 | Cyclooxygenase-1 (COX-1) | Cyclooxygenase-2 (COX-2) | PGH2_SHEEP | PTGS2 | Prostaglandin G/H synthase (Cyclooxygenase-2) | Prostaglandin G/H synthase (cyclooxygenase)
Type:
Protein
Mol. Mass.:
68976.98
Organism:
Ovis aries (Sheep)
Description:
n/a
Residue:
603
Sequence:
MLARALLLCAAVVCGAANPCCSHPCQNRGVCMSVGFDQYKCDCTRTGFYGENCTTPEFLTRIKLLLKPTPDTVHYILTHFKGVWNIVNKISFLRNMIMRYVLTSRSHLIESPPTYNVHYSYKSWEAFSNLSYYTRALPPVPDDCPTPMGVKGRKELPDSKEVVKKVLLRRKFIPDPQGTNLMFAFFAQHFTHQFFKTDIERGPAFTKGKNHGVDLSHVYGESLERQHNRRLFKDGKMKYQMINGEMYPPTVKDTQVEMIYPPHIPEHLKFAVGQEVFGLVPGLMMYATIWLREHNRVCDVLKQEHPEWGDEQLFQTSRLILIGETIKIVIEDYVQHLSGYHFKLKFDPELLFNQQFQYQNRIAAEFNTLYHWHPLLPDVFQIDGQEYNYQQFIYNNSVLLEHGVTQFVESFTRQIAGRVAGRRNLPAAVEKVSKASLDQSREMKYQSFNEYRKRFLLKPYESFEELTGEKEMAAELEALYGDIDAMELYPALLVEKPAPDAIFGETMVEAGAPFSLKGLMGNPICSPEYWKPSTFGGEVGFKIINTASIQSLICSNVKGCPFTSFSVQDAHLTKTVTINASSSHSGLDDINPTVLLKERSTEL
Inhibitor
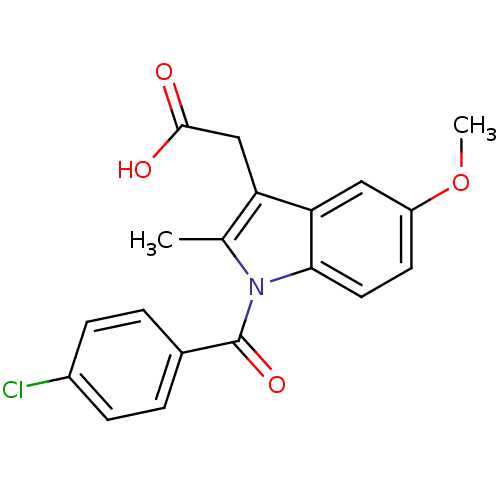
Name:
BDBM17638
Synonyms:
2-{1-[(4-chlorophenyl)carbonyl]-5-methoxy-2-methyl-1H-indol-3-yl}acetic acid | CHEMBL6 | Indocin | Indomethacin | US11478464, Compound Indomethacin | US11786535, Compound Indomethacin | US9271961, Indomethacin | indometacin
Type:
Small organic molecule
Emp. Form.:
C19H16ClNO4
Mol. Mass.:
357.788
SMILES:
COc1ccc2n(C(=O)c3ccc(Cl)cc3)c(C)c(CC(O)=O)c2c1