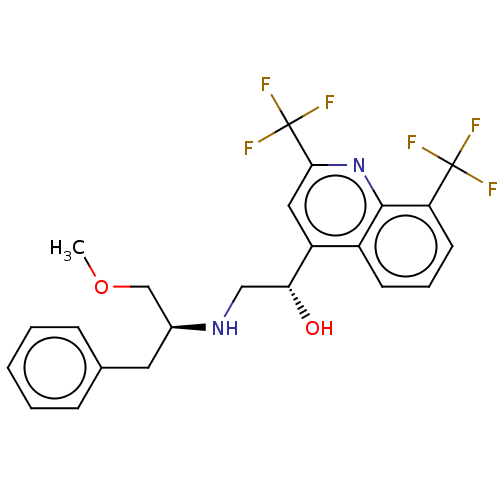
BDBM50602549 CHEMBL5183354
SMILES COC[C@H](Cc1ccccc1)NC[C@@H](O)c1cc(nc2c(cccc12)C(F)(F)F)C(F)(F)F
InChI Key InChIKey=TZXAENYIJDOHQI-UHFFFAOYSA-N
Data 5 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50602549
Found 5 hits for monomerid = 50602549
Affinity DataIC50: 2.30E+3nMAssay Description:Inhibition of CYP3A4 in human liver microsomes assessed as reduction in 6-beta-hydroxy-testosterone formation using testosterone as substrate preincu...More data for this Ligand-Target Pair
Affinity DataIC50: 2.80E+3nMAssay Description:Inhibition of CYP3A4 in human liver microsomes assessed as reduction in 1'-hydroxy midazolam formation using midazolam as substrate preincubated for ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.90E+3nMAssay Description:Inhibition of CYP3A4 in human liver microsomes assessed as reduction in 6-beta-hydroxy-testosterone formation using testosterone as substrate preincu...More data for this Ligand-Target Pair
Affinity DataIC50: 5.90E+3nMAssay Description:Inhibition of CYP3A4 in human liver microsomes assessed as reduction in 1'-hydroxy midazolam formation using midazolam as substrate preincubated for ...More data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
University of Picardy Jules Verne
Curated by ChEMBL
University of Picardy Jules Verne
Curated by ChEMBL
Affinity DataIC50: 1.73E+4nMAssay Description:Inhibition of hERG channel expressed in CHO cells at holding potential of -80 mV by whole cell patch clamp methodMore data for this Ligand-Target Pair