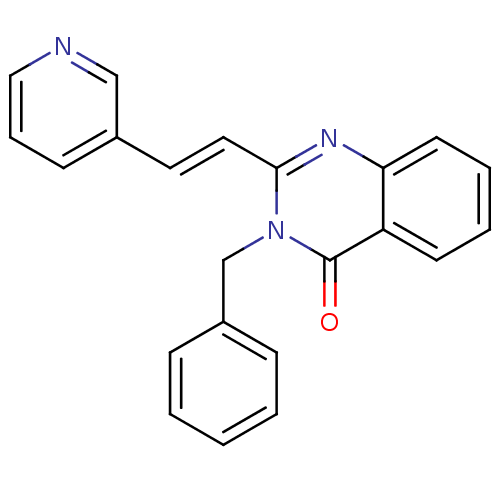
BDBM48804 3-(phenylmethyl)-2-[(E)-2-(3-pyridinyl)ethenyl]-4-quinazolinone::3-(phenylmethyl)-2-[(E)-2-pyridin-3-ylethenyl]quinazolin-4-one::3-benzyl-2-[(E)-2-(3-pyridyl)vinyl]quinazolin-4-one::3-benzyl-2-[(E)-2-pyridin-3-ylethenyl]quinazolin-4-one::MLS000709026::SMR000289793::cid_5738263
SMILES O=c1n(Cc2ccccc2)c(\C=C\c2cccnc2)nc2ccccc12
InChI Key InChIKey=GEKDQXSPTHHANP-OUKQBFOZSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 13 hits for monomerid = 48804
Found 13 hits for monomerid = 48804
TargetEukaryotic translation initiation factor 4 gamma 1(Homo sapiens (Human))
Emory University
Curated by PubChem BioAssay
Emory University
Curated by PubChem BioAssay
Affinity DataIC50: 5.77E+3nMAssay Description:Dose Response Confirmation for Small Molecule Inhibitors of Eukaryotic Translation Initiation NIH Molecular Libraries Screening Centers Network [MLSC...More data for this Ligand-Target Pair
Affinity DataIC50: 4.25E+4nMAssay Description:Project Title: A screen for modulators of human Rad51, a key DNA repair protein Application Number: MH084119 Assay Submitter: Dr. Alex Mazin Submitte...More data for this Ligand-Target Pair
Affinity DataIC50: 3.70E+4nMAssay Description:Project Title: A screen for modulators of human Rad51, a key DNA repair protein Application Number: MH084119 Assay Submitter: Dr. Alex Mazin Submitte...More data for this Ligand-Target Pair
TargetHexokinase HKDC1 [W721R](Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 3.20E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetHexokinase HKDC1 [W721R](Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 8.00E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
Affinity DataIC50: 3.50E+4nMAssay Description:Inhibition of human RAD51-mediated DNA strand exchange at using pBSK (+) gapped and linear dsDNA substrates by joint molecule formation assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.74E+4nMAssay Description:Inhibition of RAD51 (unknown origin) using 32P-labeled 90 mer ssDNA as substrate assessed as reduction in DNA D-loop formation preincubated for 30 mi...More data for this Ligand-Target Pair
TargetCell cycle checkpoint protein RAD1(Homo sapiens (Human))
Drexel University College Of Medicine
Curated by ChEMBL
Drexel University College Of Medicine
Curated by ChEMBL
Affinity DataEC50: 2.70E+4nMAssay Description:Inhibition of RAD1 (unknown origin) binding to ssDNAMore data for this Ligand-Target Pair
TargetCell cycle checkpoint protein RAD1(Homo sapiens (Human))
Drexel University College Of Medicine
Curated by ChEMBL
Drexel University College Of Medicine
Curated by ChEMBL
Affinity DataKd: 5.60E+3nMAssay Description:Binding affinity to RAD1 (unknown origin) by surface plasmon resonance methodMore data for this Ligand-Target Pair
Affinity DataIC50: 3.50E+4nMAssay Description:Inhibition of human RAD51-mediated DNA branch migration using [32P]-labeled 5'-joint molecules after 8 hrs by branch migration assayMore data for this Ligand-Target Pair
Affinity DataIC50: 6.00E+3nMAssay Description:Inhibition of human RAD51More data for this Ligand-Target Pair
Affinity DataIC50: 2.74E+4nMAssay Description:Inhibition of human RAD51 using 90-mer ssDNA as substrate measured after 30 mins by D-loop assayMore data for this Ligand-Target Pair
Affinity DataKd: 5.60E+3nMAssay Description:Binding affinity to human RAD51 in absence of ATP by SPR methodMore data for this Ligand-Target Pair