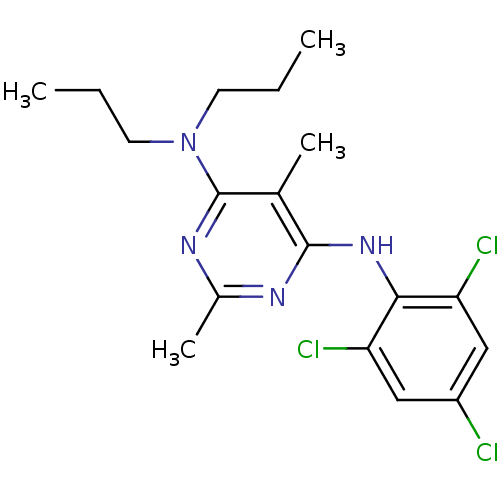
Affinity DataKi: 0.000650nM ΔG°: -72.4kJ/mole IC50: 4.5nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.00550nM ΔG°: -66.9kJ/mole IC50: 21nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.00700nM IC50: 5nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.0180nM ΔG°: -63.8kJ/mole IC50: 5.30nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.120nM ΔG°: -58.9kJ/mole IC50: 15nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.140nM IC50: 5nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.150nM IC50: 7.5nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.180nM ΔG°: -57.9kJ/mole IC50: 48nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.200nM ΔG°: -57.6kJ/mole IC50: 3.5nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.200nM ΔG°: -57.6kJ/mole IC50: 29nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.230nM IC50: 3nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
TargetSerine protease hepsin(Homo sapiens (Human))
Universidade Federal De Pernambuco
Curated by ChEMBL
Universidade Federal De Pernambuco
Curated by ChEMBL
Affinity DataKi: 0.270nMAssay Description:Inhibition of recombinant human hepsin preincubated for 30 mins followed by Boc-QLR-AMC substrate addition by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.300nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
Affinity DataKi: 0.300nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
Affinity DataKi: 0.340nM IC50: 38nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.360nM ΔG°: -56.1kJ/mole IC50: 29nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.370nM IC50: 7nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.460nM ΔG°: -55.4kJ/mole IC50: 34nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.580nM IC50: 60nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
TargetSerine protease hepsin(Homo sapiens (Human))
Universidade Federal De Pernambuco
Curated by ChEMBL
Universidade Federal De Pernambuco
Curated by ChEMBL
Affinity DataKi: 0.670nMAssay Description:Inhibition of recombinant human hepsin preincubated for 30 mins followed by Boc-QLR-AMC substrate addition by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.780nM IC50: 110nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 0.794nMAssay Description:Displacement of 3H-oxytocin from human recombinant oxytocin receptor expressed in CHO cells by filtration binding assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.990nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 1.10nM ΔG°: -53.2kJ/mole IC50: 11nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
Affinity DataKi: 1.30nM IC50: 38nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
TargetSuppressor of tumorigenicity 14 protein(Homo sapiens (Human))
Universidade Federal De Pernambuco
Curated by ChEMBL
Universidade Federal De Pernambuco
Curated by ChEMBL
Affinity DataKi: 1.30nMAssay Description:Inhibition of recombinant human matriptase preincubated for 30 mins followed by Boc-QLR-AMC substrate addition by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nM IC50: 56nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 1.5nM IC50: 95nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
Affinity DataKi: 1.58nMAssay Description:Binding affinity to human recombinant oxytocin receptor expressed in CHO cells by fluorescence polarization assayMore data for this Ligand-Target Pair
Affinity DataKi: 1.60nM IC50: 51nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
TargetCorticotropin-releasing factor receptor 1(Homo sapiens (Human))
Neurocrine Biosciences
Curated by ChEMBL
Neurocrine Biosciences
Curated by ChEMBL
Affinity DataKi: 1.70nMAssay Description:Inhibition of [125I]-CRF binding to human Corticotropin releasing factor receptor 1 tested at 6-12 dosesMore data for this Ligand-Target Pair
Affinity DataKi: 1.80nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
Affinity DataKi: 1.80nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
Affinity DataKi: 1.80nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
Affinity DataKi: 2nM IC50: 45nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:Antagonist activity at human recombinant oxytocin receptor expressed in CHO cells by FLIPR assayMore data for this Ligand-Target Pair
TargetCorticotropin-releasing factor receptor 1(Homo sapiens (Human))
Neurocrine Biosciences
Curated by ChEMBL
Neurocrine Biosciences
Curated by ChEMBL
Affinity DataKi: 2nMAssay Description:Inhibition of [125I]-CRF binding to human Corticotropin releasing factor receptor 1 tested at 6-12 dosesMore data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
Affinity DataKi: 2.10nM ΔG°: -51.5kJ/mole IC50: 15nMpH: 7.4 T: 2°CAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
Affinity DataKi: 2.10nM IC50: 19nMAssay Description:Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a...More data for this Ligand-Target Pair
TargetCorticotropin-releasing factor receptor 1(Homo sapiens (Human))
Neurocrine Biosciences
Curated by ChEMBL
Neurocrine Biosciences
Curated by ChEMBL
Affinity DataKi: 2.30nMAssay Description:Inhibition of [125I]-CRF binding to human Corticotropin releasing factor receptor 1 tested at 6-12 dosesMore data for this Ligand-Target Pair
TargetChymase(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 2.30nMAssay Description:Inhibition of human skin chymaseMore data for this Ligand-Target Pair
Affinity DataKi: 2.40nMAssay Description:Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp...More data for this Ligand-Target Pair
TargetCorticotropin-releasing factor receptor 1(Homo sapiens (Human))
Neurocrine Biosciences
Curated by ChEMBL
Neurocrine Biosciences
Curated by ChEMBL
Affinity DataKi: 2.5nMAssay Description:Inhibition of [125I]-CRF binding to human Corticotropin releasing factor receptor 1 tested at 6-12 dosesMore data for this Ligand-Target Pair
TargetSuppressor of tumorigenicity 14 protein(Homo sapiens (Human))
Universidade Federal De Pernambuco
Curated by ChEMBL
Universidade Federal De Pernambuco
Curated by ChEMBL
Affinity DataKi: 2.5nMAssay Description:Inhibition of recombinant human matriptase preincubated for 30 mins followed by Boc-QLR-AMC substrate addition by fluorescence assayMore data for this Ligand-Target Pair

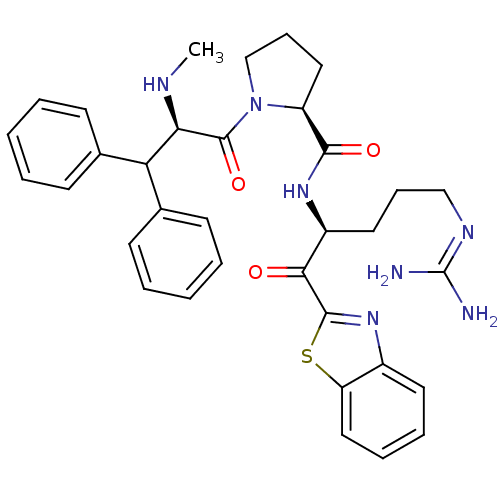

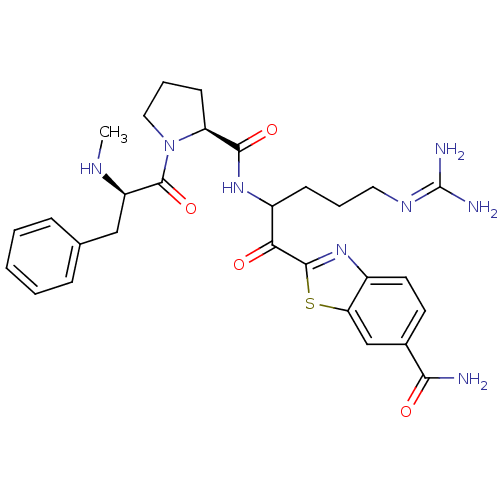
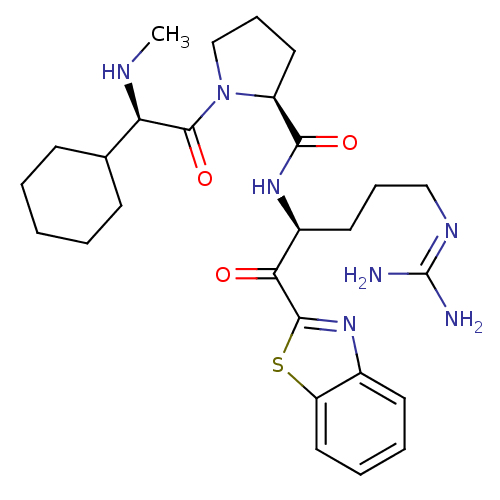
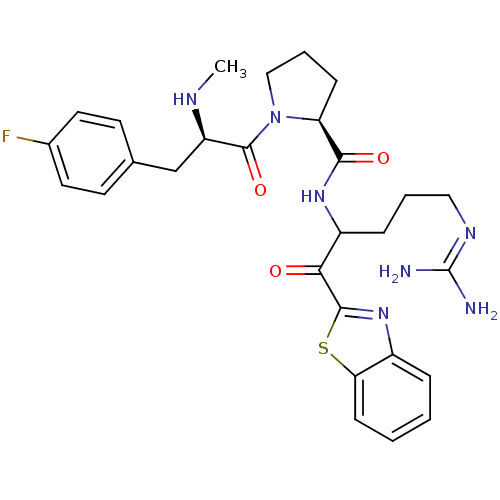
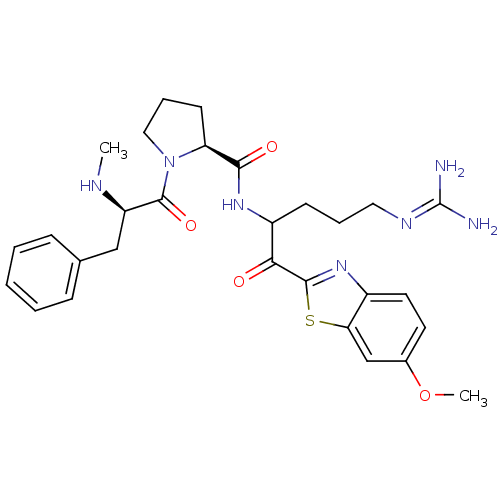

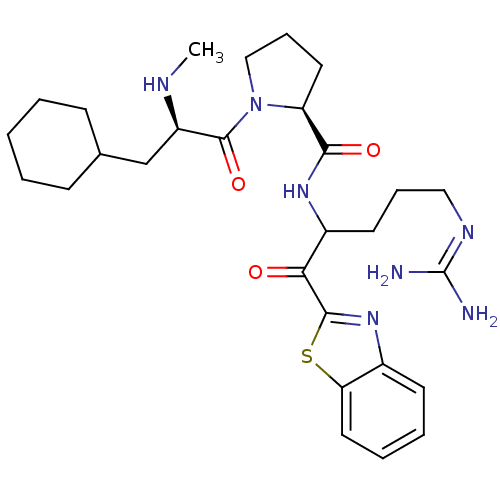
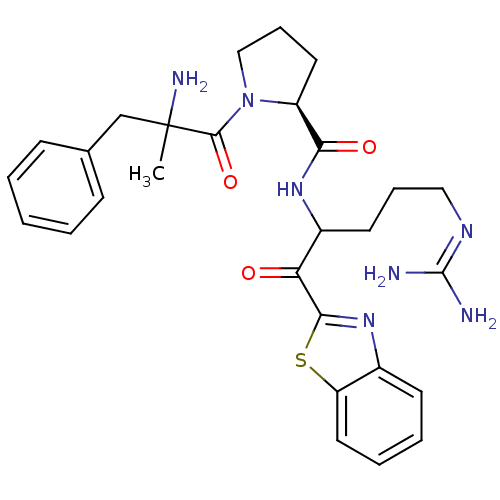

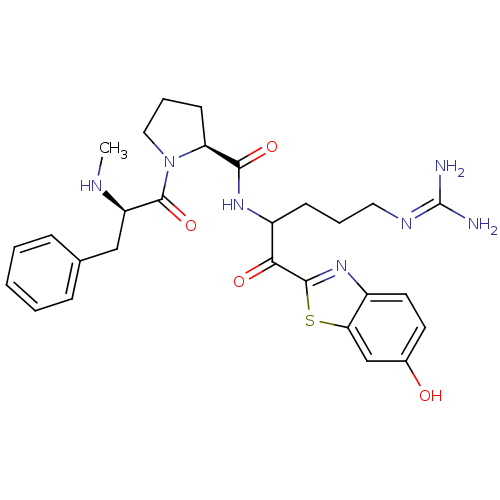


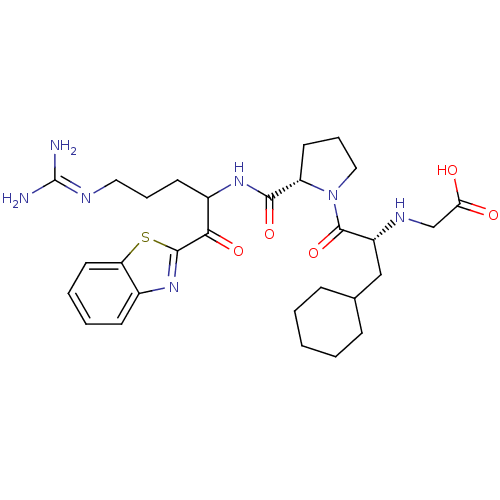
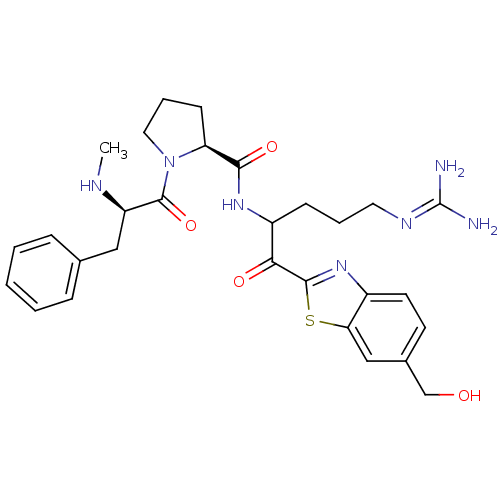
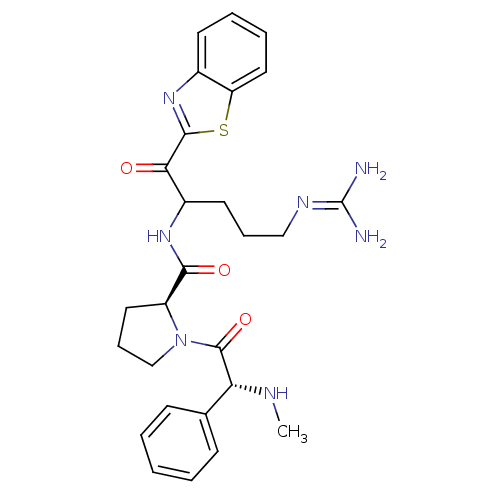



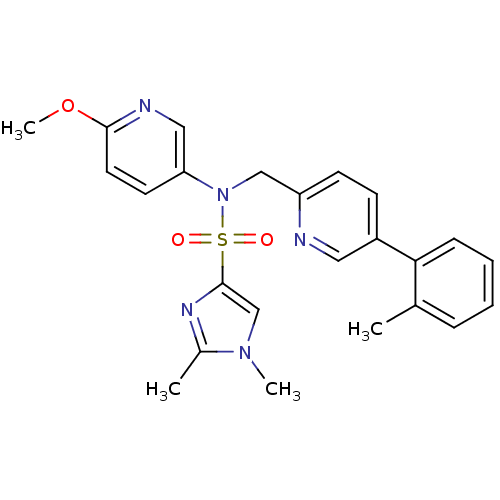

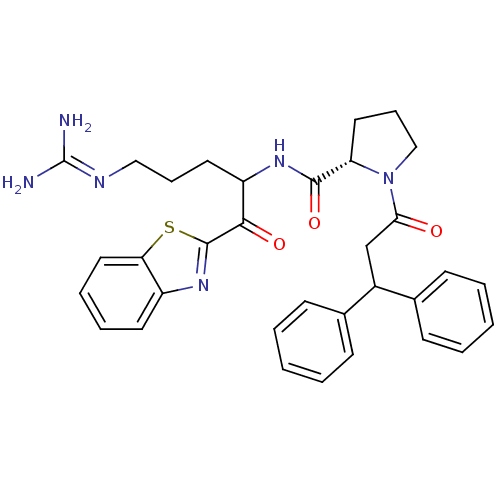
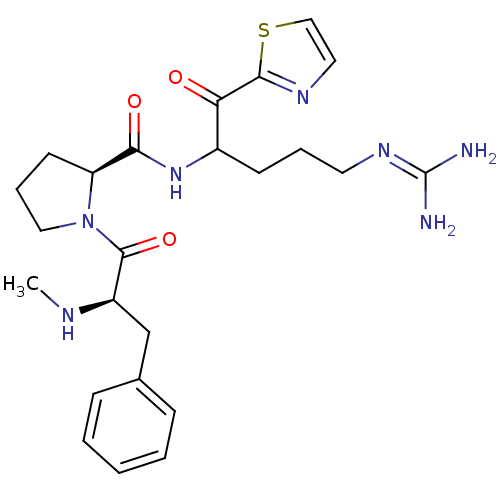
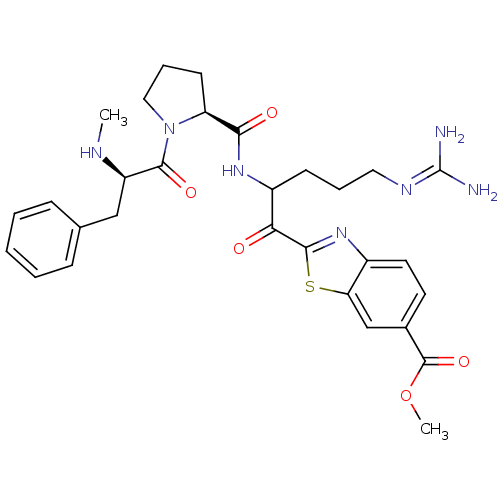
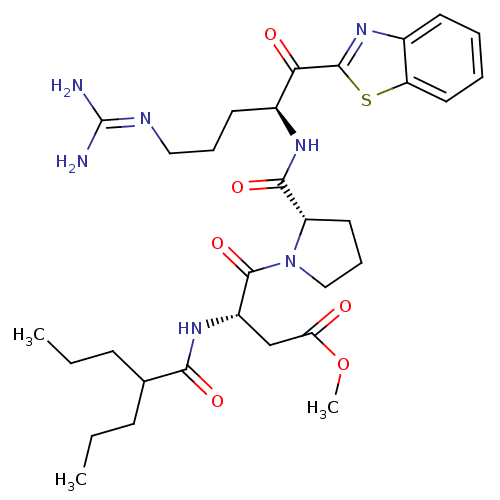

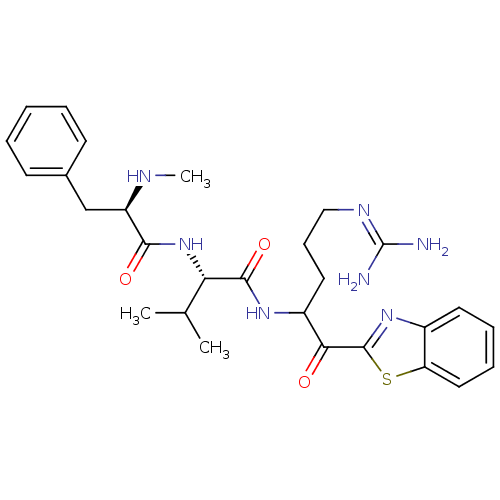

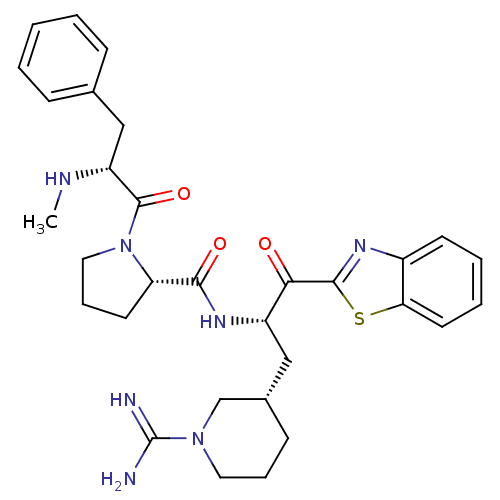

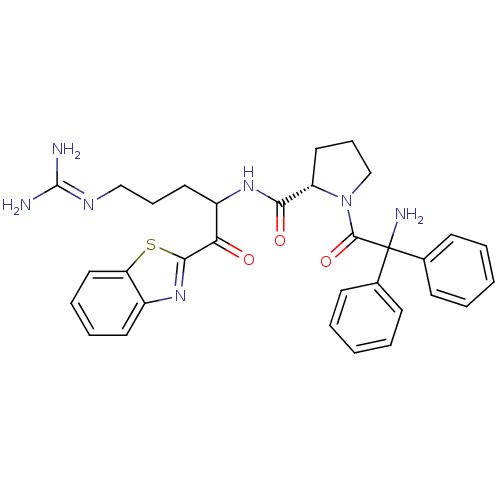
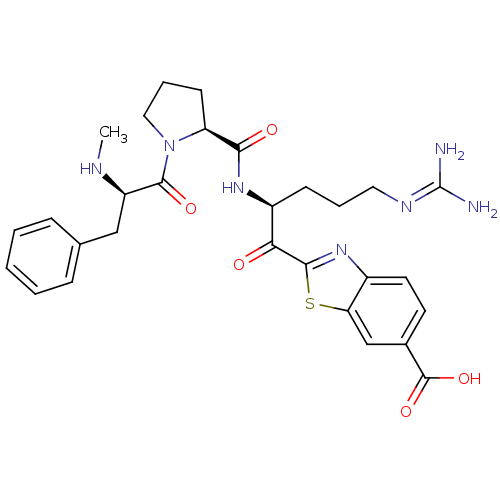
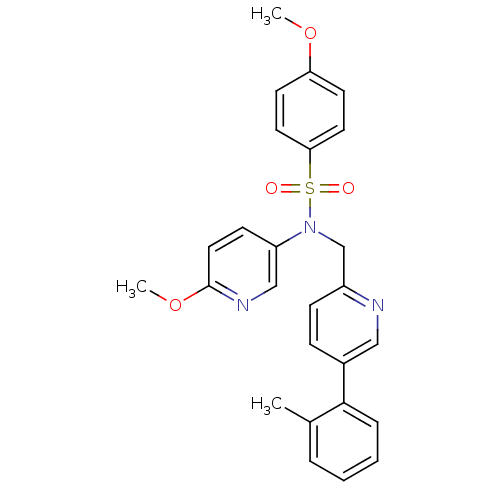

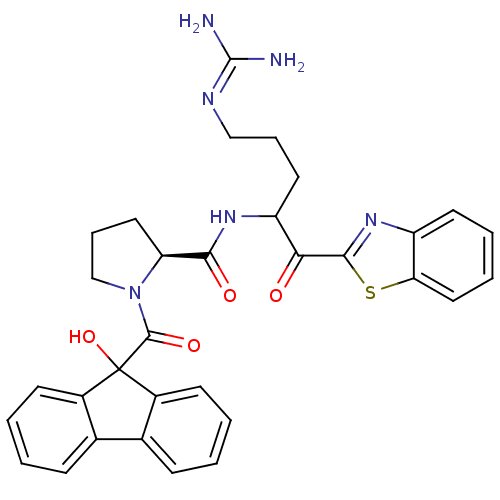

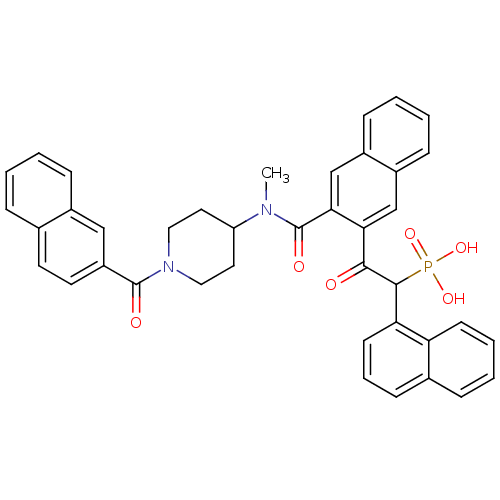
 3D Structure (crystal)
3D Structure (crystal)