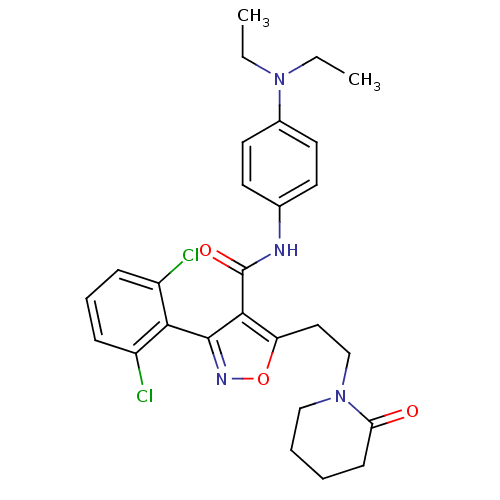
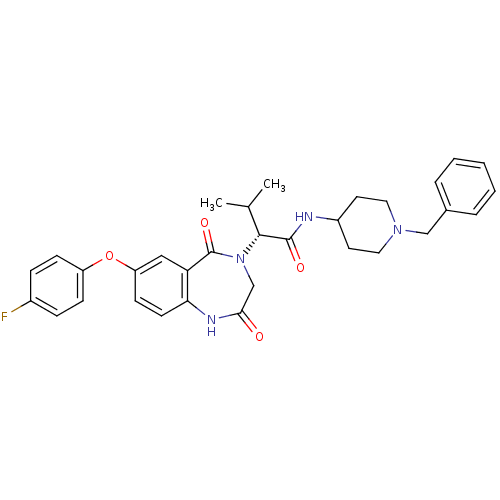
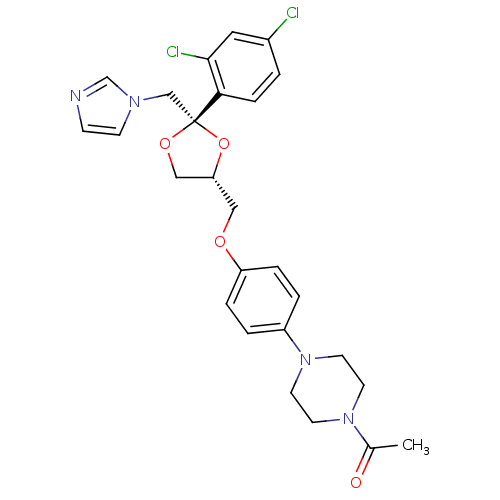
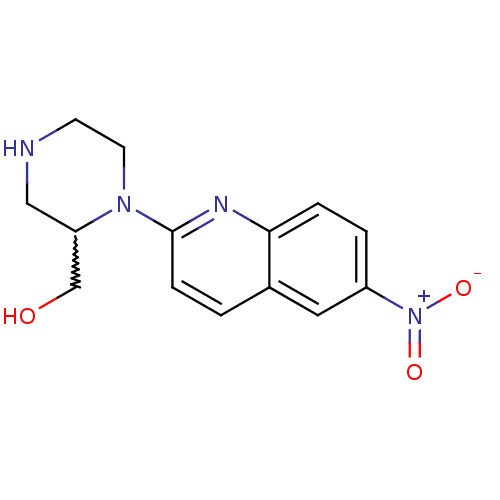
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Inha University
Curated by ChEMBL
Inha University
Curated by ChEMBL
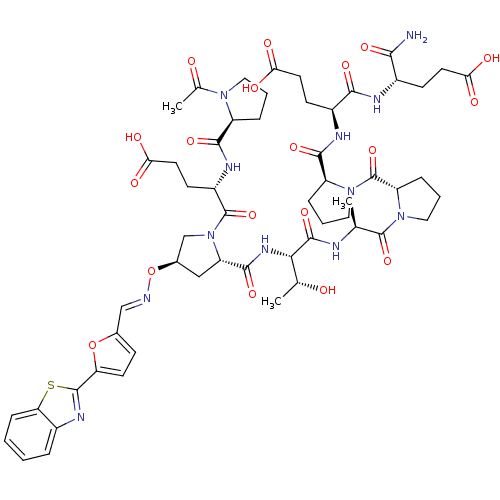
Affinity DataKi: 0.240nMAssay Description:Displacement of [3H]citalopram from Sprague-Dawley rat SERTMore data for this Ligand-Target Pair
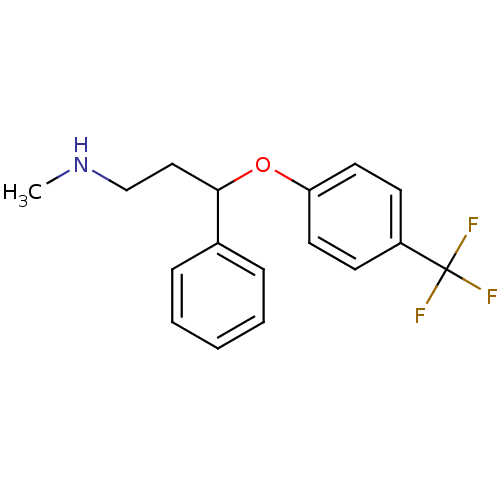
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Inha University
Curated by ChEMBL
Inha University
Curated by ChEMBL
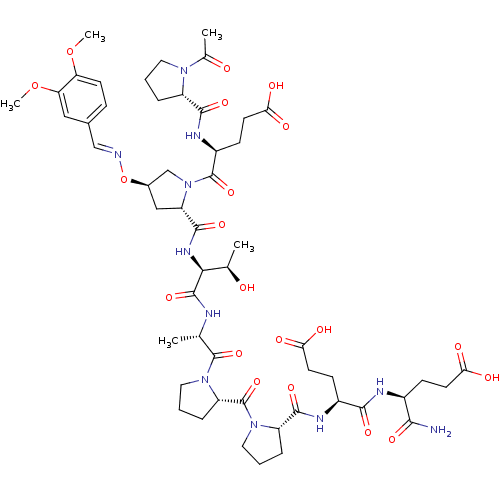
Affinity DataKi: 0.430nMAssay Description:Displacement of [3H]citalopram from Sprague-Dawley rat SERTMore data for this Ligand-Target Pair
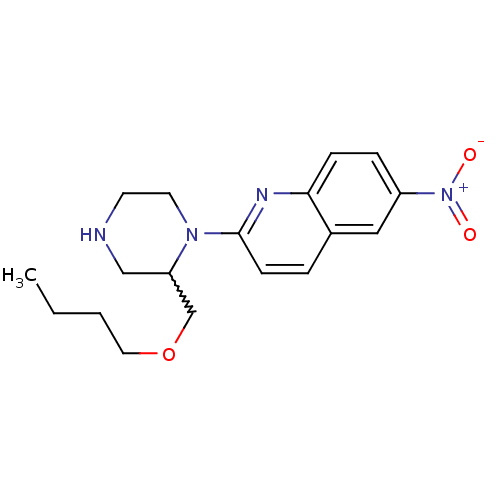
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Inha University
Curated by ChEMBL
Inha University
Curated by ChEMBL
Affinity DataKi: 0.430nMAssay Description:Displacement of [3H]citalopram from Sprague-Dawley rat SERTMore data for this Ligand-Target Pair
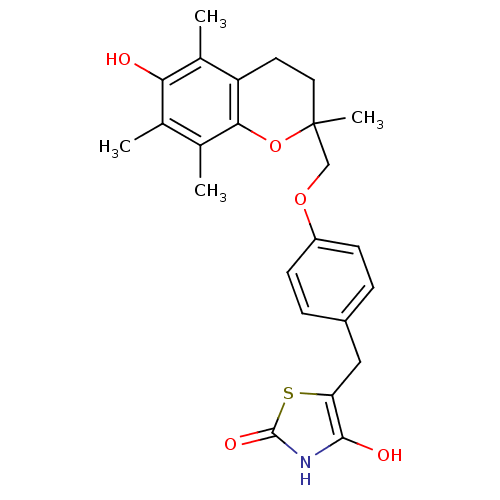
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Inha University
Curated by ChEMBL
Inha University
Curated by ChEMBL
Affinity DataKi: 0.680nMAssay Description:Displacement of [3H]citalopram from Sprague-Dawley rat SERTMore data for this Ligand-Target Pair
Affinity DataKi: 4.40nMAssay Description:Displacement of [125I]Ghrelin from human GHSR1a receptorMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Inha University
Curated by ChEMBL
Inha University
Curated by ChEMBL
Affinity DataKi: 5.67nMAssay Description:Displacement of [3H]citalopram from Sprague-Dawley rat SERTMore data for this Ligand-Target Pair
Affinity DataKi: 7.80nMAssay Description:Compound was tested for inhibitory activity against carboxypeptidase AMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Inha University
Curated by ChEMBL
Inha University
Curated by ChEMBL
Affinity DataKi: 8.70nMAssay Description:Displacement of [3H]citalopram from Sprague-Dawley rat SERTMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(Rattus norvegicus (rat))
Inha University
Curated by ChEMBL
Inha University
Curated by ChEMBL
Affinity DataKi: 15.4nMAssay Description:Displacement of [3H]citalopram from Sprague-Dawley rat SERTMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Homo sapiens (Human))
Seoul National University
Curated by ChEMBL
Seoul National University
Curated by ChEMBL
Affinity DataKi: 400nMAssay Description:Binding affinity to human recombinant PPARgamma by Cheng-Prusoff equation based competitive binding TR-FRET assayMore data for this Ligand-Target Pair
Affinity DataKi: 2.60E+3nMAssay Description:Displacement of FITC-conjugated (S)-4-((S)-1-acetylpyrrolidine-2-carboxamido)-5-((2S,4R)-2-((2S,3R)-1-((S)-1-((S)-2-((S)-2-((S)-1-((S)-1-amino-4-carb...More data for this Ligand-Target Pair
Affinity DataKi: 2.70E+3nMAssay Description:Displacement of FITC-conjugated (S)-4-((S)-1-acetylpyrrolidine-2-carboxamido)-5-((2S,4R)-2-((2S,3R)-1-((S)-1-((S)-2-((S)-2-((S)-1-((S)-1-amino-4-carb...More data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor alpha(Homo sapiens (Human))
Seoul National University
Curated by ChEMBL
Seoul National University
Curated by ChEMBL
Affinity DataKi: 4.50E+3nMAssay Description:Binding affinity to human recombinant PPARalpha by Cheng-Prusoff equation based competitive binding TR-FRET assayMore data for this Ligand-Target Pair
Affinity DataKi: 5.40E+3nMAssay Description:Displacement of FITC-conjugated (S)-4-((S)-1-acetylpyrrolidine-2-carboxamido)-5-((2S,4R)-2-((2S,3R)-1-((S)-1-((S)-2-((S)-2-((S)-1-((S)-1-amino-4-carb...More data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Homo sapiens (Human))
Seoul National University
Curated by ChEMBL
Seoul National University
Curated by ChEMBL
Affinity DataKi: 6.70E+3nMAssay Description:Binding affinity to human recombinant PPARgamma by Cheng-Prusoff equation based competitive binding TR-FRET assayMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Homo sapiens (Human))
Seoul National University
Curated by ChEMBL
Seoul National University
Curated by ChEMBL
Affinity DataKi: 7.00E+3nMAssay Description:Binding affinity to human recombinant PPARgamma by Cheng-Prusoff equation based competitive binding TR-FRET assayMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor alpha(Homo sapiens (Human))
Seoul National University
Curated by ChEMBL
Seoul National University
Curated by ChEMBL
Affinity DataKi: 8.00E+3nMAssay Description:Binding affinity to human recombinant PPARalpha by Cheng-Prusoff equation based competitive binding TR-FRET assayMore data for this Ligand-Target Pair
Affinity DataKi: 8.70E+3nMAssay Description:Displacement of FITC-conjugated (S)-4-((S)-1-acetylpyrrolidine-2-carboxamido)-5-((2S,4R)-2-((2S,3R)-1-((S)-1-((S)-2-((S)-2-((S)-1-((S)-1-amino-4-carb...More data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor alpha(Homo sapiens (Human))
Seoul National University
Curated by ChEMBL
Seoul National University
Curated by ChEMBL
Affinity DataKi: 9.50E+3nMAssay Description:Binding affinity to human recombinant PPARalpha by Cheng-Prusoff equation based competitive binding TR-FRET assayMore data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 4.10nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: <6nMAssay Description:Inhibition of CYP2D6 in human liver microsomeMore data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:Antagonist activity at mouse GHSR1 receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 8nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 8nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as blockade of intracellular Ca2+ mobilizationMore data for this Ligand-Target Pair
Affinity DataIC50: 8nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 15nMAssay Description:Partial agonist activity at mouse GPR142 expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as blockade of intracellular Ca2+ mobilizationMore data for this Ligand-Target Pair
Affinity DataIC50: 16.5nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 26nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 28nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 50nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: <62nMAssay Description:Inhibition of CYP3A4 in human liver microsomeMore data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Homo sapiens (Human))
Korea Institute Of Science And Technology
Curated by ChEMBL
Korea Institute Of Science And Technology
Curated by ChEMBL
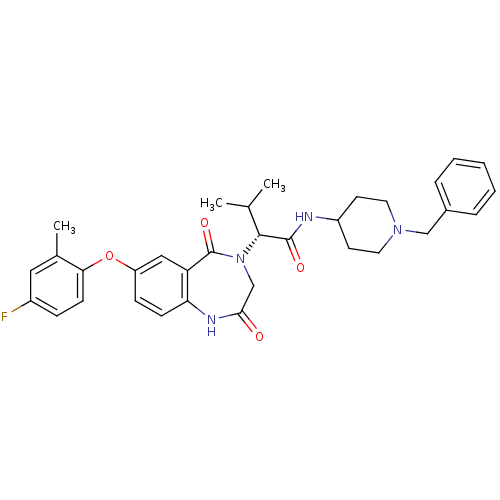
Affinity DataIC50: 63nMAssay Description:Displacement of [3H]-methylspiperone from dopamine D2L receptor (unknown origin) after 60 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 79nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Homo sapiens (Human))
Korea Institute Of Science And Technology
Curated by ChEMBL
Korea Institute Of Science And Technology
Curated by ChEMBL
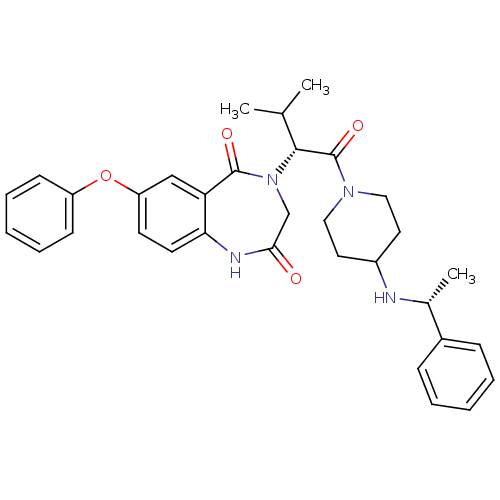
Affinity DataIC50: 86nMAssay Description:Displacement of [3H]-methylspiperone from dopamine D2L receptor (unknown origin) after 60 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 143nMAssay Description:Antagonist activity at human GHSR1a receptor assessed as intracellular Ca2+ concentration by aequorin luminescent assayMore data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Homo sapiens (Human))
Korea Institute Of Science And Technology
Curated by ChEMBL
Korea Institute Of Science And Technology
Curated by ChEMBL
Affinity DataIC50: 147nMAssay Description:Displacement of [3H]-methylspiperone from dopamine D2L receptor (unknown origin) after 60 minsMore data for this Ligand-Target Pair








 3D Structure (crystal)
3D Structure (crystal)