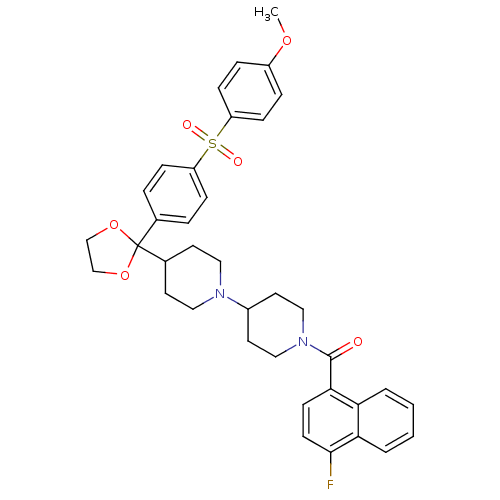
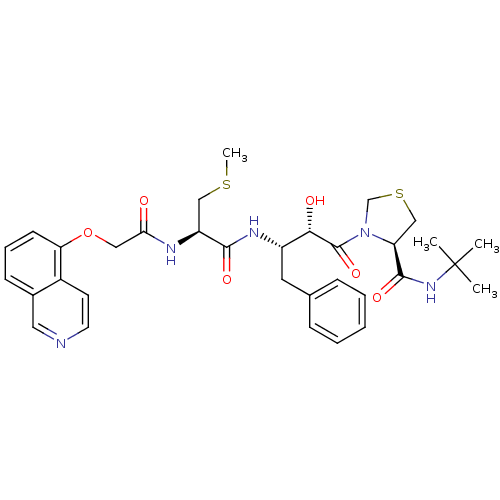
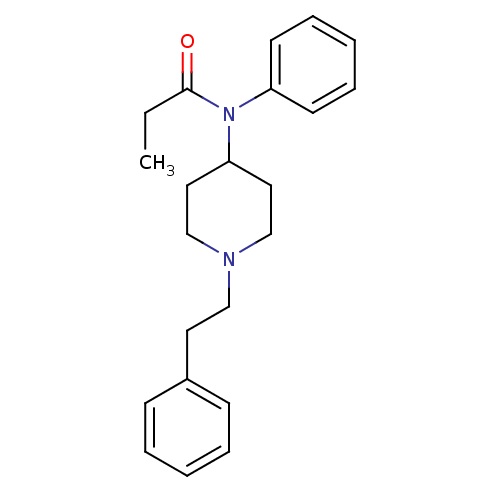
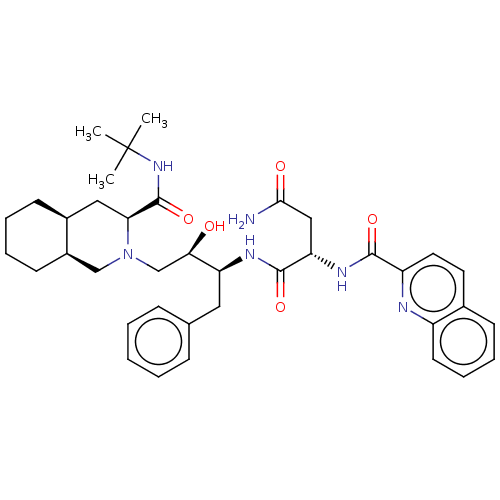
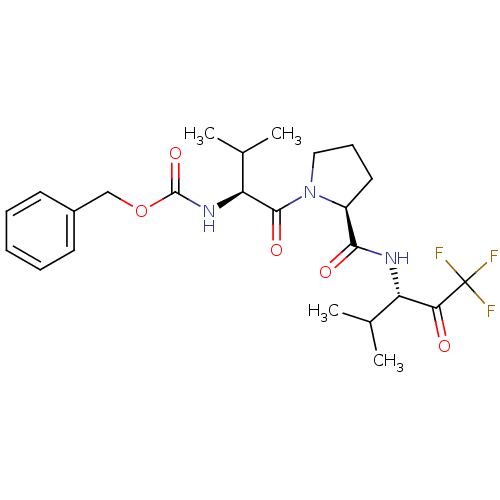
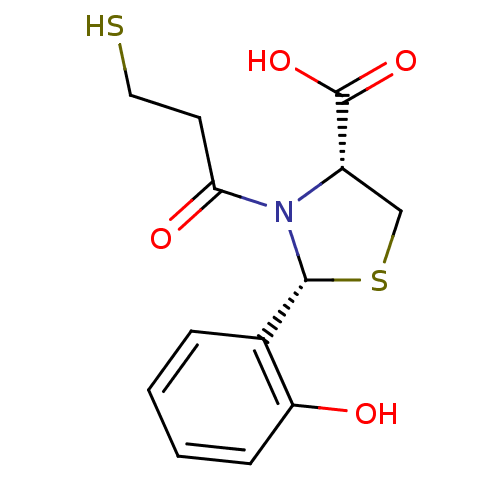
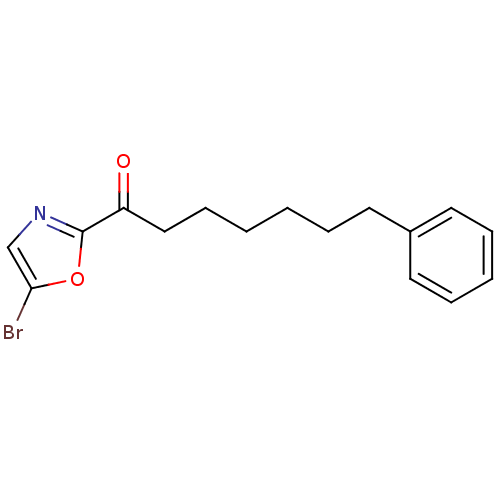
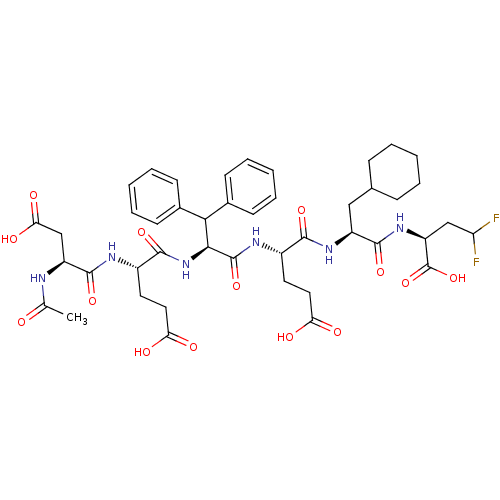
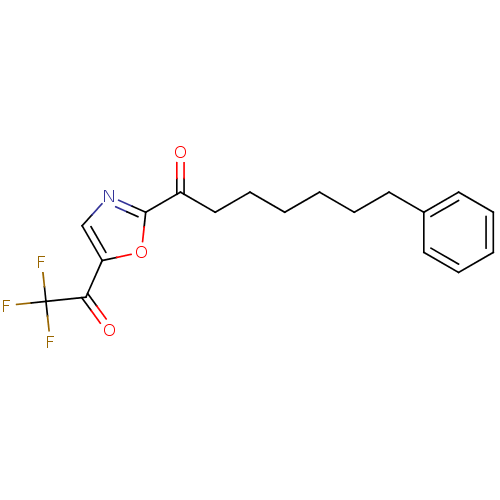
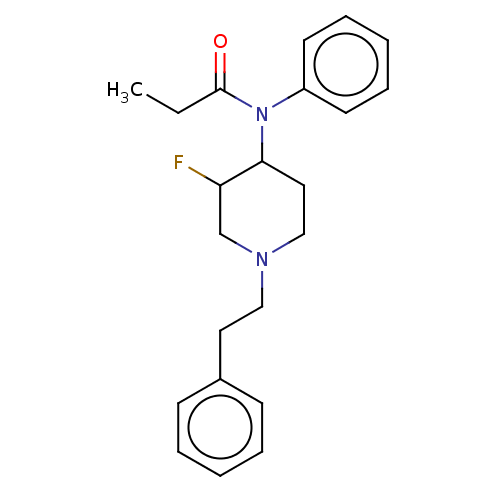
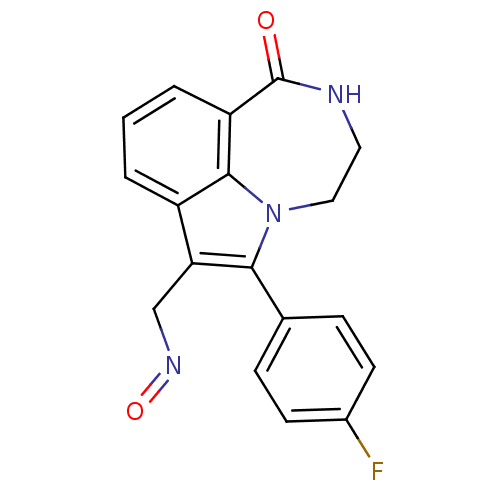
Affinity DataKi: 0.0100nMAssay Description:Antagonist activity at muscarinic M2 receptor (unknown origin) assessed as inhibition constantMore data for this Ligand-Target Pair
TargetNeutrophil elastase(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 0.0250nMAssay Description:Inhibition of human neutrophil elastaseMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.0420nMAssay Description:Inhibition of Thrombin (unknown origin)More data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.100nMAssay Description:Inhibition of Thrombin (unknown origin)More data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.270nMAssay Description:Inhibition of Thrombin (unknown origin)More data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A(Homo sapiens (Human))
Bristol-Myers Squibb
Curated by ChEMBL
Bristol-Myers Squibb
Curated by ChEMBL
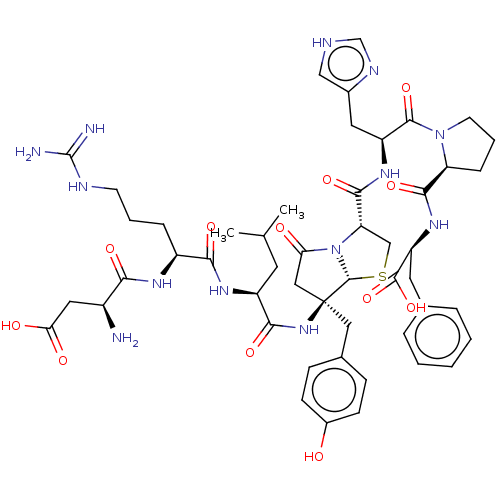
Affinity DataKi: 0.300nMAssay Description:Inhibition of His6 tagged human FKBP12 expressed in Escherichia coli BL21(DE3) cells using succinylALPF-p-nitroanilide as substrate by fluorescence p...More data for this Ligand-Target Pair
Affinity DataKi: 0.330nMAssay Description:Inhibition of HIV1 proteaseMore data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
TargetNeutrophil elastase(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 0.490nMAssay Description:Inhibition of human neutrophil elastaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.510nMAssay Description:Antagonist activity at muscarinic M2 receptor (unknown origin) assessed as inhibition constantMore data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 0.550nMAssay Description:Inhibition of human dopamine D2 receptorMore data for this Ligand-Target Pair
TargetNeutrophil elastase(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 0.600nMAssay Description:Inhibition of human neutrophil elastaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.740nMAssay Description:Inhibition of HIV1 proteaseMore data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 0.800nMAssay Description:Inhibition of Thrombin (unknown origin)More data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
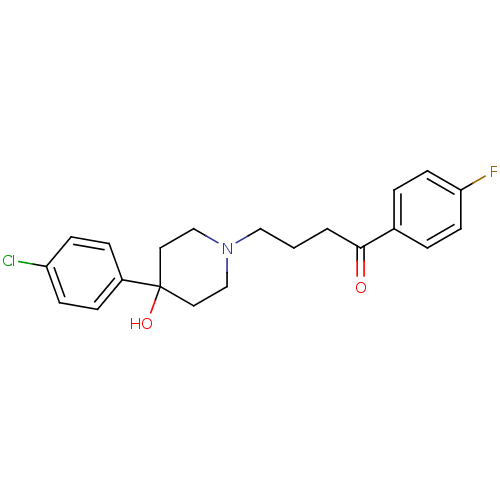
Affinity DataKi: 0.800nMAssay Description:Antagonist activity against human V1A receptor expressed in human 1321N1 cellsMore data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 1nMAssay Description:Displacement of [I125I]AngII form AT2 receptor in rat liver membrane incubated for 1.5 hrs by gamma counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Displacement of [I125I]AngII form AT1 receptor in rat liver membrane incubated for 1.5 hrs by gamma counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Binding affinity to mu opioid receptor (unknown origin) expressed in HEK cells at pH 7.4More data for this Ligand-Target Pair
TargetProthrombin(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 1.10nMAssay Description:Inhibition of Thrombin (unknown origin)More data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP3 subtype(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 1.10nMAssay Description:Inhibition of EP3 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:Displacement of [I125I]AngII form AT2 receptor in rat liver membrane incubated for 1.5 hrs by gamma counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Inhibition of HIV-1 protease assessed as inhibition constantMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Antagonist activity against human V1A receptor expressed in human 1321N1 cellsMore data for this Ligand-Target Pair
TargetNeutrophil elastase(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 1.60nMAssay Description:Inhibition of human neutrophil elastaseMore data for this Ligand-Target Pair
TargetSigma non-opioid intracellular receptor 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 1.90nMAssay Description:Inhibition of sigma 1 receptorMore data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
TargetFatty-acid amide hydrolase 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
TargetProstaglandin E2 receptor EP3 subtype(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
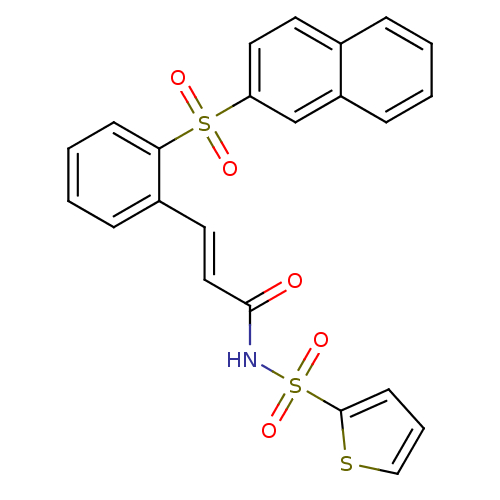
Affinity DataKi: 2.10nMAssay Description:Inhibition of EP3 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 2.20nMAssay Description:Inhibition of ACE (unknown origin)More data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 2.84nMAssay Description:Inhibition of human dopamine D2 receptorMore data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
TargetFatty-acid amide hydrolase 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 3nMAssay Description:Inhibition of Hepatitis C virus NS3/NS4A proteaseMore data for this Ligand-Target Pair
TargetNeutrophil elastase(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 3nMAssay Description:Inhibition of human neutrophil elastaseMore data for this Ligand-Target Pair
TargetSigma non-opioid intracellular receptor 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 3.40nMAssay Description:Inhibition of sigma 1 receptorMore data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
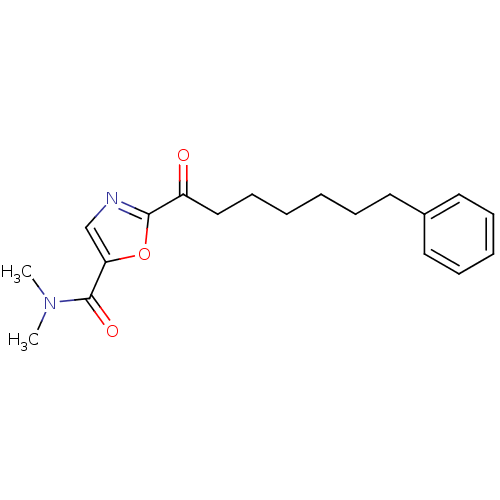
Affinity DataKi: 3.60nMAssay Description:Inhibition of human SSTR2More data for this Ligand-Target Pair
Ligand Info
Affinity DataKi: 3.70nMAssay Description:Binding affinity to mu opioid receptor (unknown origin) expressed in HEK cells at pH 6.5More data for this Ligand-Target Pair
Affinity DataKi: 3.70nMAssay Description:Displacement of [I125I]AngII form AT2 receptor in rat liver membrane incubated for 1.5 hrs by gamma counting methodMore data for this Ligand-Target Pair
TargetD(3) dopamine receptor(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 4.73nMAssay Description:Inhibition of human dopamine D3 receptorMore data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
TargetProthrombin(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataKi: 5nMAssay Description:Inhibition of human Thrombin using S-2238 as substrate assessed as release of p-nitroaniline preincubated for 240 secs followed by substrate addition...More data for this Ligand-Target Pair
Affinity DataKi: 5nMAssay Description:Antagonist activity at muscarinic M2 receptor (unknown origin) assessed as inhibition constantMore data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
TargetPoly [ADP-ribose] polymerase 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 6nMAssay Description:Inhibition of human PARP-1More data for this Ligand-Target Pair
TargetFatty-acid amide hydrolase 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
TargetPoly [ADP-ribose] polymerase 1(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 6nMAssay Description:Inhibition of human PARP-1More data for this Ligand-Target Pair
TargetProstaglandin E2 receptor EP3 subtype(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 6.70nMAssay Description:Inhibition of EP3 receptorMore data for this Ligand-Target Pair

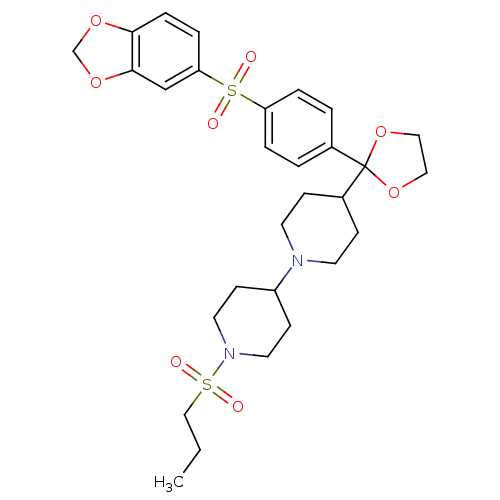


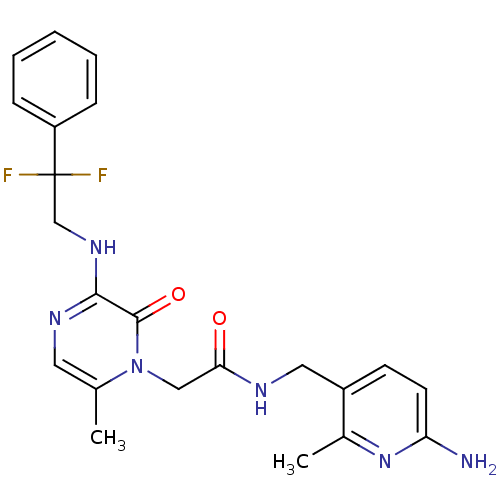


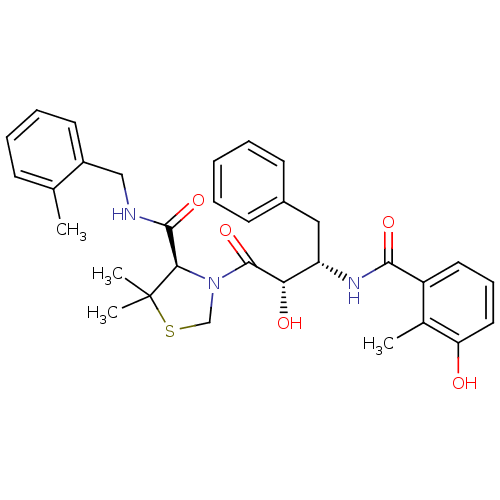
 3D Structure (crystal)
3D Structure (crystal)